All published articles of this journal are available on ScienceDirect.
Comparative Analysis of Grain Sorghum and Corn as Raw Materials for Bioethanol Production using Very High Gravity Fermentation
Abstract
Introduction
Global warming is reducing the availability of conventional feedstocks for bioethanol production, particularly corn. Therefore, grain sorghum is a viable choice because it is naturally drought-resistant and cost-effective. In Ukraine, sorghum can reduce raw material expenses by 15–20% in arid regions. The aim of this study is a comparative assessment of the technological characteristics of mash and wort from grain sorghum and corn prepared using VHG technology.
Materials and Methods
The grain sorghum hybrid “Brigga” was compared with the corn hybrid “DKS 3730”. Wort with dry matter >270 g/L was prepared via hydroenzymatic treatment. Dynamic viscosity was measured using a rotational viscometer. Fermentation was conducted using dry yeast, followed by distillation and chromatographic analysis to determine ethanol concentration and by-products.
Results
Both grains have similar starch levels (64–78%), but differ in protein and non-starch polysaccharide content. After 72 hours of fermentation, both wort types contained approximately 14% vol. ethanol, with a yield of 1.52–1.54 g/(l·h). Raw sorghum wort distillates showed 11.1% lower levels of volatile by-products than corn.
Discussion
Studies show that sorghum wort maintains a low viscosity despite higher hemicellulose and β-glucan content. This thinner consistency of sorghum mash is advantageous for VHG fermentation as it allows for higher grain loading while maintaining efficient fermentation.
Conclusion
Grain sorghum demonstrates a technological advantage over corn in bioethanol production using VHG technology. Understanding these approaches will help achieve post-fermentation ethanol concentrations exceeding 18% v/v, improving the economic efficiency of biofuel production.
1. INTRODUCTION
Since the beginning of the new millennium, bioethanol has taken a prominent place among liquid fuels. As a result, significant attention is being devoted to expanding the raw material base and improving production technologies. At present, much research is focused on developing technologies for ethanol production from inexpensive cellulosic substrates [1]; however, economically viable biofuel technologies utilising lignocellulose face numerous challenges [2]. This stimulates further research aimed at increasing the energy efficiency and environmental sustainability of bioethanol production using readily available sugar- and starch-containing grain raw materials and their by-products [3–9]. As for raw materials, corn is the most widely used for bioethanol production [10, 11]. However, global warming and reduced precipitation, as manifestations of climate change, require the search for resilient grain crops suitable for regions of risky agriculture. In areas with poor agronomic conditions, grain sorghum can act as an alternative to corn and improve farm economic stability [12]. Its high yield and drought resistance make this energy crop a promising raw material for biofuel production [13]. Cultivating energy crops on low-productive and marginal lands unsuitable for traditional crop farming enables the production of biofuels that meet sustainability criteria in accordance with EU legislation [14].
Grain sorghum is an annual herbaceous plant capable of producing yields even under unfavourable conditions, up to 4-6 t of grain per hectare, with a starch content of up to 75% [6, 7].
To date, the issue of identifying alternative raw materials for bioethanol production in Ukraine remains insufficiently studied, yet it is highly relevant given the growing demand for fuels from renewable sources. At the same time, it is necessary to investigate the technological aspects of using sorghum for bioethanol production to implement modifications in the technological process, its equipment configuration, and to assess potential production costs in comparison with corn.
Sorghum starch is similar to corn starch, but some structural differences should be taken into consideration when producing bioethanol. Corn contains about 70–75% amylopectin and 25–30% amylose. The level of amylose in sorghum depends on the variety, usually from 12 to 28%, so it has more amylopectin. There are also some soluble (0.7–4.2%) and reducing sugars (0.05–0.53%), and this can affect the onset of fermentation [9, 15, 16].
The structure of starch influences its susceptibility to breakdown, with amylopectin being branched and therefore readily degraded by α-amylase, whereas amylose forms tight helices that are more resistant to enzymatic breakdown. Consequently, starch with a higher amylopectin content is easier to liquefy and ferment, reducing the amount of glucoamylase required. Alternatively, when processing starch with a high amylose content, it is advisable to enhance the enzymatic complex by adding glucoamylase [16].
The cell walls of corn grain contain β-glucans (≈ 0.1% of the grain), arabinoxylans, and phenolic acids, which significantly affect the rheological properties of the mash and can inhibit enzymatic hydrolysis, particularly α-amylase and glucoamylase. This necessitates the addition of β-glucanase and xylanase to reduce mash viscosity and improve enzyme access to starch. Sorghum grain is characterized by a relatively low β-glucan content (0.06–0.43%), although certain amounts of arabinoxylans are also present within the cell wall matrix. However, the overall viscosity of sorghum mash is lower; therefore, the primary enzymatic supplement for sorghum should be xylanase, while β-glucanase is required to a much lesser extent [17, 18]. Corn also contains antinutritional compounds such as phytates, α-amylase inhibitors, and protease inhibitors, which can reduce the efficiency of enzymatic degradation of natural polymers [15].
Corn protein is primarily composed of zein, which accounts for approximately 60% of the total protein content. In sorghum, the main protein is prolamin, making up approximately 48–70% of the total grain protein. The protein barrier surrounding starch granules hinders amylolytic enzymes from hydrolysing starch [19].
Corn grain contains up to 6% total lipids (mostly in the germ), while the endosperm contains about 1% [20]. An elevated fat content in processing by-products (Distillers Dried Grains with Solubles, DDGS) can reduce quality due to the susceptibility of lipids (fats) to rancidity and the shortening of shelf life. At the same time, sorghum DDGS contains about 10.5% fat, which is lower than in corn, where the lipid fraction is significantly higher. This gives sorghum DDGS lower susceptibility to oxidation and, consequently, more stable storage properties [21].
Studies comparing sorghum and corn for bioethanol production have already been conducted, but most focused on ethanol-associated by-product content in raw distillates [12], leaving the specific features of using sorghum in Very High Gravity (VHG) technology unclear. VHG involves an elevated concentration of dry matter in the nutrient wort at the start of fermentation (>270 g/L) [3]. At present, the most common method for converting starchy polysaccharides into ethanol is Simultaneous Saccharification and Fermentation (SSF). This method alone allows reducing the initial osmotic stress on yeast at high initial sugar concentrations, as may occur when using the SHF method [22].
The higher amylopectin content in corn grain promotes faster mash liquefaction during hydrolysis, whereas sorghum contains more phenolic compounds, which may inhibit enzymatic processes. To optimise the SSF process, it is advisable to use enzyme preparations: xylanase and β-glucanase to reduce the viscosity of corn mash, and phytase to improve the availability of phosphorus-containing compounds in sorghum. This was supported by the results of Pérez Carrillo et al. and other researchers, who demonstrated that the use of enzyme combinations significantly reduced viscosity caused by water-soluble polysaccharides compared to the application of each enzyme individually [15].
There is a lack of studies on the viscosity of VHG wort from sorghum, which is potentially lower than that of corn wort due to its lower content of β-glucans and pentosans [18]. This allows for increased dry matter and ethanol concentrations, as well as reduced energy and auxiliary material consumption during the technological process. However, a problem of physiological stress may still arise, caused by high initial sugar levels or elevated final ethanol concentrations [3]. In this context, many studies focus on determining the need for substances that can mitigate the negative effects of high wort component concentrations on yeast [3, 8, 22, 23]. There are also examples of achieving ethanol concentrations up to 20.25% v/v through fermentation of sugarcane juice using VHG technology [8]. When making VHG wort from sugar cane, the liquid becomes very thick. However, the high viscosity of such worts inhibited the fermentation kinetics due to restricted mass transfer and reduced diffusion rates of nutrients and metabolites.
The aim of this study is a comparative assessment of the technological characteristics of mash and wort from grain sorghum and corn, prepared with a dry matter content ≈28%, at the lower limit of using VHG technology, with a focus on viscosity, enzymatic efficiency, and the content of volatile by-products that affect the quality of raw distillate. Special attention was given to evaluating dynamic viscosity during hydroenzymatic treatment and SSF fermentation, which is a critical factor for the implementation of VHG processes. The obtained data will allow us to outline ways to continue research for the application of VHG technology to sorghum raw materials to obtain wort with a dry matter concentration >30% and and ethanol accumulation in the fermented wort >17% v/v.
2. MATERIALS AND METHODS
2.1. Raw Material for Mash Preparation
An early-maturing grain sorghum hybrid, the “Brigga” variety (RAGT Semences, France), was selected for comparison with the traditional raw material, corn of the hybrid variety “DKS 3730” (Monsanto, USA). Both crops are high-yielding and intended for ethanol production. The raw materials for the study were provided by the agroholding VITAGRO. According to the manufacturers, the thousand-kernel weight is 350 grams for the corn variety and 38 grams for the sorghum variety. The raw materials were milled using an LZM-1 grain mill (Ukraine) to obtain a uniform grind capable of passing through a 1 mm mesh sieve. Sieve analysis of the obtained flour confirmed a consistent particle size distribution, with over 90% of the particles for both feedstocks ranging between 0.2 mm and 0.8 mm. This level of fineness ensures a high specific surface area, facilitating optimal enzymatic access to the starch granules during the subsequent liquefaction and saccharification stages.
Starch content was determined using a hydrochloric acid solution according to ISO 10520:1997 [24]. Crude protein content was calculated using a factor of 6.25 after determining total nitrogen by the Kjeldahl method according to AOAC 979.09 [25]. Starch and protein contents in the grain were expressed on a dry matter basis. Dry matter content was determined by drying the wort on ADS-50 moisture balances (Kern, UK).
2.2. Preparation of Enzymatic Wort
For the bioconversion of starch-containing raw materials using the SSF scheme, 1.96 kg of milled material was mixed with water at a ratio of 1:2. The process was carried out in a MicroFerm laboratory fermenter (USA) with a working volume of 6 L wort. The dry matter content for all wort samples was 28 ± 0.1%. The initial pH was adjusted within the range of 4.8–6.0 and measured using an AZ-86505 laboratory analyser. The initial wort pH was 5.39 ± 0.01 for sorghum and 5.45 ± 0.01 for corn. To ensure a precise dry matter concentration of 28% across all batches, the volume of added water was meticulously adjusted based on the initial moisture content of the grain (13.8% for corn and 12.5% for sorghum), ensuring that the final weight-to-weight ratio accounted for the total solids in each specific sample.
The main set of enzymes for implementing an efficient bioethanol production process included Spezyme HN, Distillase XP, and Optimash TBG (produced by International Flavors & Fragrances, USA):
- Spezyme HN contains α-amylase and thermostable phytase, which prevents the negative impact of phytates on α-amylase activity. It also releases free phosphorus during liquefaction, which is necessary for yeast nutrition.
- Distillase XP contains glucoamylase, used for saccharification and fermentation. It includes a mixture of enzymes with optimised activity and dosing: glucoamylase, acidic α-amylase, protease, and trehalase.
- Optimash TBG is a thermostable enzyme with hydrolytic activity on non-starch polysaccharides such as cellulose, hemicellulose, β-D-glucans, and arabinoxylans.
Each wort was liquefied by adding Spezyme HN (10 U/g DM) and Optimash TBG (1.2 U/g DM). The wort was then heated to a temperature above 60 °C, with periodic stirring to prevent clumping of dry matter. Upon reaching 80 °C, the wort was liquefied, after which the temperature was further increased to 87–88 °C and maintained for 3 hours under continuous stirring. Iodine was used to check whether saccharification was complete. The absence of a blue-violet colouration during the iodine test indicated that the α-amylase had achieved complete starch hydrolysis.
2.3. Fermentation of Wort for Bioethanol Production
After the thermofermentation stage was completed, the Simultaneous Saccharification and Fermentation (SSF) stage started. The Distillase XP enzyme mixture was added to the cooled wort and then inoculated with Thermosacc ML yeast (from Denmark). The yeast contained approximately 2.0 × 1010 cells per gram. These yeasts were selected because they can withstand high temperatures, varying pH levels, and other stress conditions encountered during fermentation.
The wort was cooled and kept at a temperature of 33 ± 1°C. Distillase XP (0.35 U/g DM), the antiseptic “Bactrilon” (0.6 g/100 L wort), and orthophosphoric acid (1.2 mL/L wort) were then added to the wort. All wort samples were fermented for 72 hours in a laboratory fermenter.
2.4. Analysis of Fermented Wort
The density of the mash and wort was measured using a Rotavisc hi-vi viscometer (from IKA-Werke GmbH & Co. KG, Germany). The readings were taken every hour for 4 hours. Spezyme HN, an enzyme that breaks down starch into dextrins, was added to reduce the mash density. After fermentation, the acidity of the wort was measured by titration with NaOH under phenolphthalein control. The results are shown in grams of acid per litre of wort. To determine the ethanol content, 100 ml of distilled water was added to 200 ml of fermented wort, and the mixture was placed in a 500 ml flask with subsequent distillation to obtain 200 ml of distillate. The ethanol content in this distillate was determined using an Anton Paar DMA 4101 densimeter (Anton Paar GmbH, Austria). Bioethanol production yield was calculated as the ratio of ethanol concentration (g/L) to the duration of yeast fermentation.
The concentration of volatile by-products associated with ethanol in the fermented wort distillates was determined by gas chromatography. Standard ethanol impurities (Standard EN 15721) were analysed using an Agilent Technologies 7890B chromatograph (USA) equipped with an FID detector and HP-INNOWax capillary columns.
2.5. Statistical Analysis
All experiments were performed in triplicate, and the results were expressed as the arithmetic mean with standard deviation. Statistical analysis of the experimental data was carried out using Microsoft Office Excel (Microsoft Corporation, USA) and Origin (OriginLab Corporation, USA). The significance of differences between parameters was evaluated using pairwise multiple comparisons in Student’s t-test at a p ≤ 0.05 significance level [26].
3. RESULTS
3.1. Raw Material Characteristics
Sorghum and corn grains contain 10–15% protein and 66–66.5% starch (Fig. 1). The moisture content of the raw materials ranged from 13–14%. When expressed on a dry-matter basis, the starch content can reach up to 75%, a characteristic of high-yielding hybrids.
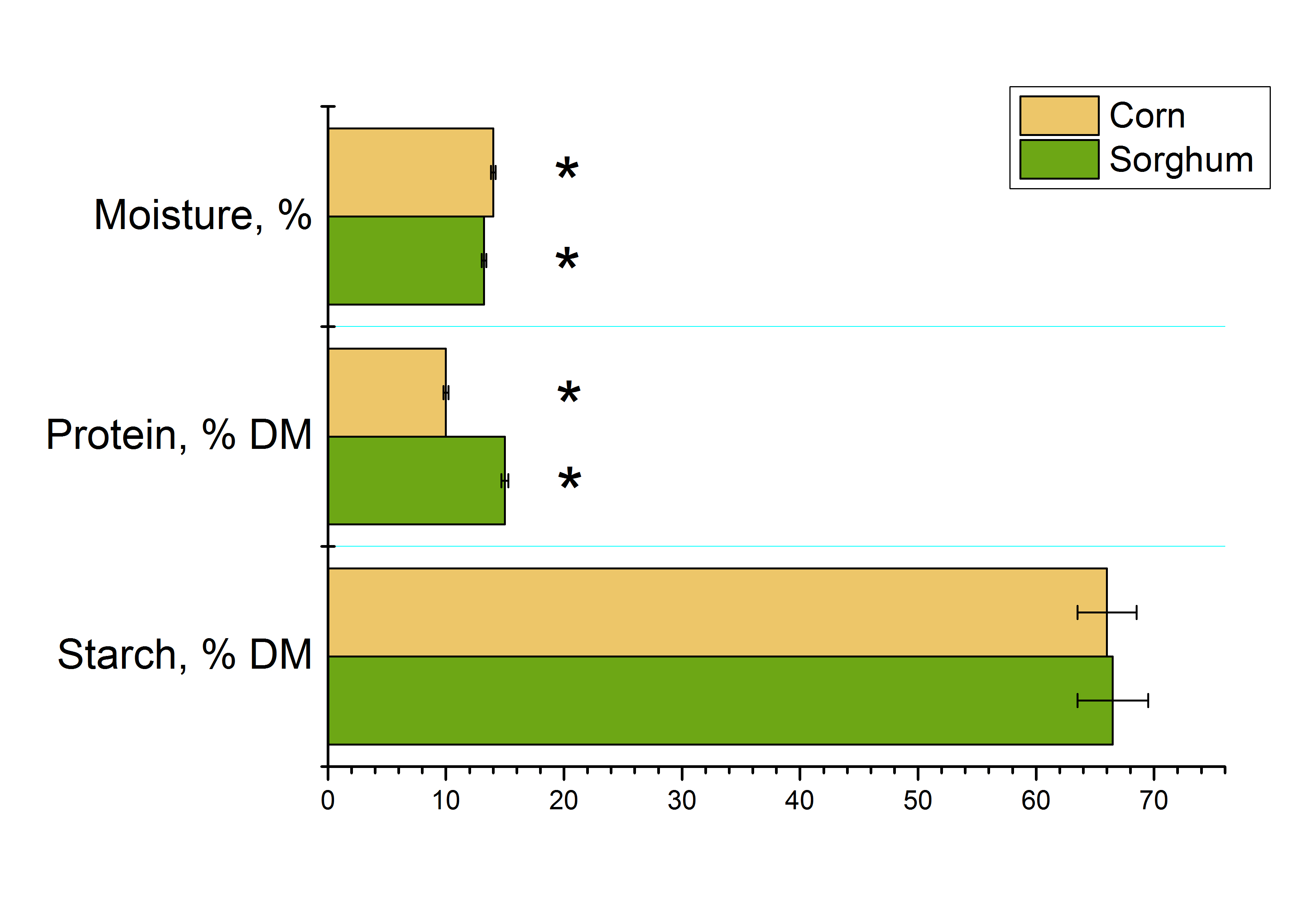
Content of the main components in sorghum and corn seed.
Note: * - data for sorghum and corn varieties are statistically different, p ≤ 0.05.
During the hydroenzymatic treatment stage, changes in dynamic viscosity were measured for sorghum and corn (Fig. 2).
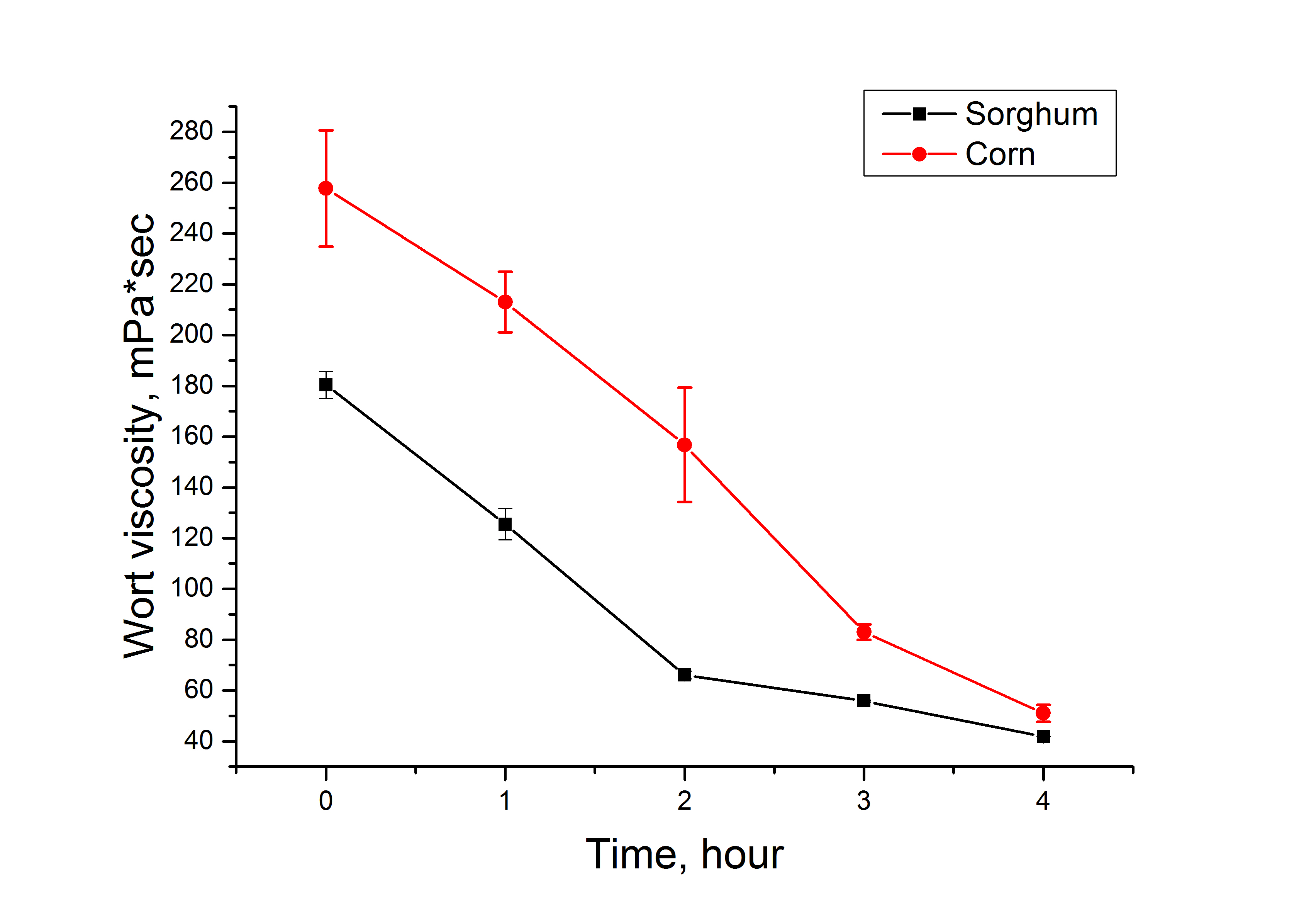
Study of the dynamics of wort viscosity before and during the liquefaction and saccharification stage.
Note: data for sorghum and corn media are statistically different, p ≤ 0.05.
During the hydroenzymatic treatment stage, mash viscosity ranged from 40 to 250 mPa·s at a rotation speed of 90 RPM. Maintaining the viscosity within the optimal range was critical, as excessive viscosity hindered efficient mixing and uniform enzyme distribution, thereby reducing the fermentation rate. The maximum allowable viscosity is 300 mPa·s. If it exceeds this value, the mash cannot flow well through the pipes, and the enzymes and other substances can not be distributed evenly. As a result, the starch will not be processed as quickly as it should. Corn mash is thicker than sorghum mash due to its different grain structure. Following the liquefaction process, both corn and sorghum mashes exhibited a significant reduction in viscosity and reached stable values within a 4-hour period. Sorghum mash exhibited a more rapid reduction in viscosity, which was attributed to its lower concentrations of β-glucans and arabinoxylans compared to corn.
The pH values of the sorghum and corn mash after enzyme treatment were almost identical, remaining in the range of 5.3 - 5.5. This means that fermentation started under similar conditions for both types of mash, and there was no need to change the pH.
3.2. Evaluation of Fermented Wort
We tested how thick the wort became during fermentation in the lab. This density, called dynamic viscosity at a rotation speed of 90 RPM, indicates how easily the mixture flows and changes with the proportion of dissolved solids in the wort (Fig. 3). The more solids in the wort, the thicker it becomes as the starch and water molecules start to interact more. This can make fermentation more difficult.
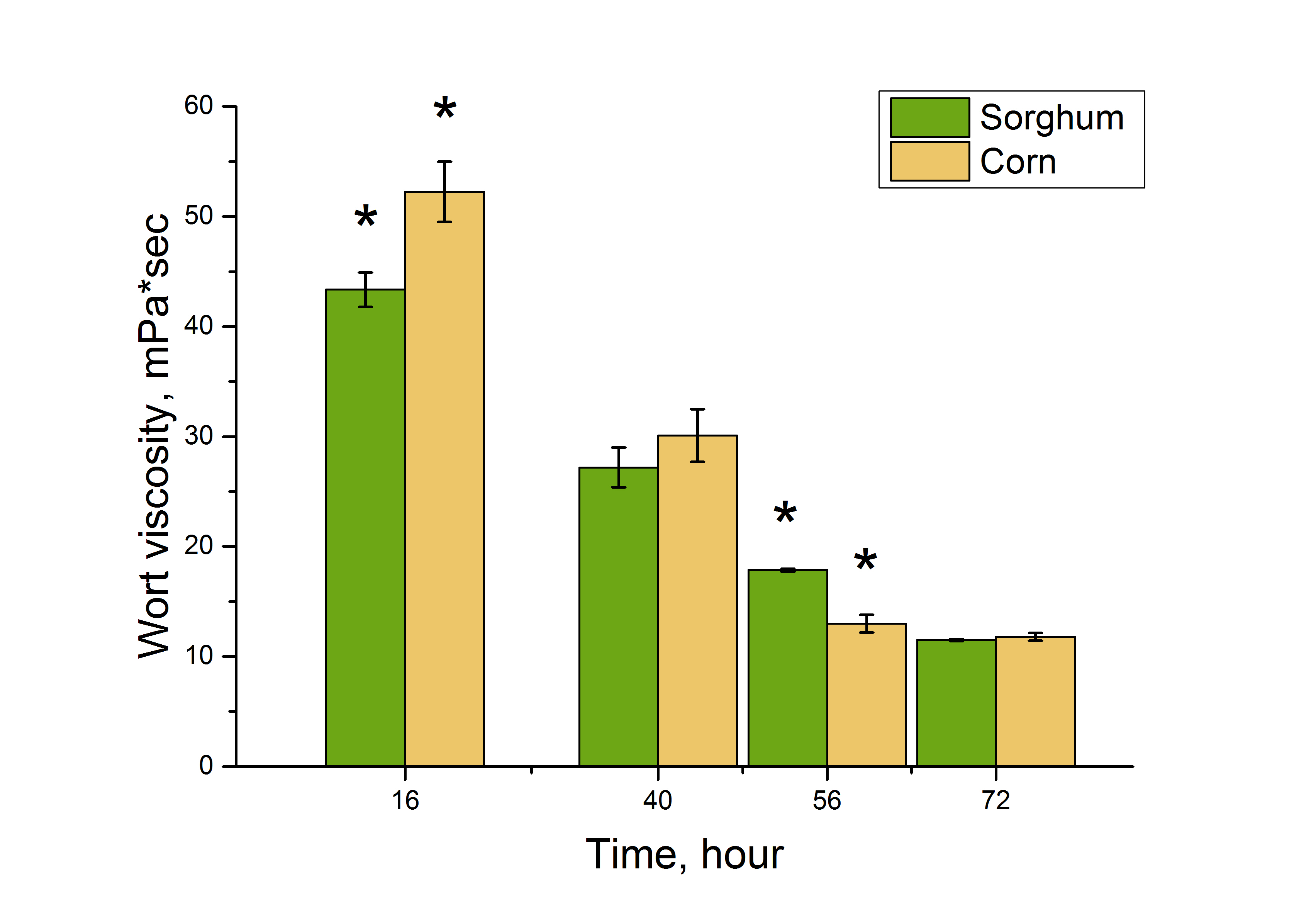
Change in dynamic viscosity during fermentation.
Note: * - data for sorghum and corn wort are statistically different, p ≤ 0.05.
At the initial stages of fermentation (16 hours), wort viscosity values were at their highest. Subsequently, a gradual decrease in viscosity was observed, indicating effective breakdown of the wort’s polymeric components. By the 56th hour of fermentation, wort viscosity had decreased significantly. By the 72nd hour, the fermentation process had stabilised. In both cases, wort viscosity reached nearly the same values, confirming the completion of the main stages of wort fermentation.
3.3. Ethanol Accumulation
To evaluate ethanol accumulation in the fermented wort, fermented sorghum and corn samples were obtained. The results of the fermented wort analysis are presented in Table 1.
| Raw Material | Acidity, g/L | Alcohol Content, % | Bioethanol Production Yield, g/(L·h) |
|---|---|---|---|
| Sorghum | 0.41 ± 0.05 a | 13.90 ± 0.43 a | 1.52 ± 0.05 a |
| Corn | 0.55 ± 0.04 b | 14.04 ± 0.18 a | 1.54 ± 0.02 a |
Note: Values with different letters (a, b) within a column are statistically different, p ≤ 0.05.
Acidity in corn mash was 26% higher than in sorghum mash. This increased acidity in corn mash is primarily attributed to its chemical composition, specifically a higher concentration of organic acids and phosphorus compounds compared to sorghum. Crucially, the observed acidity levels remained within the optimal range for yeast activity, and no signs of bacterial contamination were detected, indicating that the difference did not negatively impact the overall fermentation efficiency. Ethanol content in sorghum and corn mash after fermentation did not differ significantly.
The concentration of volatile by-products (VB) associated with ethanol in the distillates of corn and sorghum mash is presented in Table 2. Overall, raw sorghum wort distillates showed 11.1% lower levels of volatile by-products. While the contents of higher alcohols and ketones did not differ significantly between corn and sorghum mash, the levels of aldehydes, esters, and methanol were 19%, 45%, and 15% lower, respectively, in sorghum mash.
| Volatile By-Products (VB) | VB Content in Raw Corn Distillate, mg/dm³ of 100% Ethanol | VB Content in Raw Sorghum Distillate, mg/dm³ of 100% Ethanol, mg/dm³ of 100% Ethanol |
|---|---|---|
| Higher alcohols | 6230.6 ± 442.2 a | 5678.8 ± 425.8 a |
| Аldehydes | 331.6 ± 24.9 a | 268.3 ± 18.9 b |
| Еsters | 343.3 ± 25.7 a | 189.0 ± 13.9 b |
| Меthanol | 33.8 ± 2.3 a | 28.6 ± 2.0 b |
| Кеtons | 9.3 ± 0.7 a | 9.6 ± 0.7 a |
| Total VB | 6948.6 ± 443.7 | 6174.3 ± 426.5 |
Note: Values with different letters (a, b) within a row are statistically different, p ≤ 0.05.
4. DISCUSSION
The data we obtained on the protein and starch content in sorghum and corn grains correspond to the ranges given in the literature [5,12,27,28]. Bioethanol yield from grain largely depends on the content and degree of enzymatic degradation of starch and protein [13]. Corn kernels contain, on a dry matter basis, 61–78% starch, about 10% other polysaccharides, 6–12% protein, and 3–6% lipids [27]. The starch and protein contents in the samples of corn “DKS 3730” and sorghum “Brigga” correspond to these ranges (Fig. 1). Grain sorghum has a starch content similar to that of corn, ranging from 64–74% of dry matter [13]. The protein content in the selected sorghum variety is near the upper limit of reported ranges (8–15% dry matter) found in the literature [13, 28].
Experimental studies have shown that ethanol yield increases as the final wort viscosity decreases [29]. Elevated viscosity in mashes is typically caused by the presence of hemicelluloses and β-glucans [18]. However, the concentration of these non-starch polysaccharides is notably lower in sorghum than in corn, which significantly influenced the rheological properties of the wort. In this study, the dynamic viscosity was quantified to assess its impact on fermentation performance. Monitoring this parameter ensured that the dynamic viscosity remained consistently below the critical technological threshold of 300 mPa·s, thereby preventing mass transfer limitations.
Corn mash and wort exhibit higher viscosity at the initial stages but liquefy more rapidly during fermentation, whereas sorghum shows a more gradual decrease in viscosity (Figs. 2 and 3). At the same time, the lower initial viscosity of sorghum mash allows preparation of media with higher dry matter concentrations without excessive thickening. Thus, in the context of VHG technology, sorghum permits higher wort concentrations.
This observation highlights the interplay between the structural and chemical differences of the two crops. While sorghum’s higher amylopectin content facilitates starch accessibility and liquefaction, these advantages are balanced by the presence of phenolic compounds, which may act as enzyme inhibitors. In contrast, corn lacks such phenolic inhibition but presents higher initial viscosity due to its starch architecture. Our results suggest that at a 28% dry matter concentration, the enhanced liquefaction efficiency of sorghum’s amylopectin compensates for the inhibitory effects of its polyphenols, resulting in a final ethanol yield comparable to that of corn.
The obtained results indicate that differences in the chemical composition of corn and sorghum affect the rheological properties of the wort and fermentation efficiency. The concentration of volatile by-products in raw distillates from sorghum is slightly lower than that from corn, but the differences are not significant for the quality of fuel ethanol.
Ethanol concentration in the final product for both types of raw material, at a dry matter concentration of 28%, did not differ statistically (Table 1). After 72 hours of fermentation, mash from both crops reached an ethanol concentration of 14% v/v. Previous studies on VHG co-fermentation of sorghum grain and juice achieved 16.8% ethanol [15]; however, this required a significantly longer time (96 hours) and a much higher initial wort concentration (starch-to-water ratio of 1:2, or 34% dry matter in the mash).
Some studies primarily analysed the effect of the hydromodule on the content of volatile by-products in raw distillate [12], without focusing on achieving high ethanol yield. In these studies, bioethanol yield often reached only 50–70% of the theoretical value, indicating incomplete utilisation of raw material. For the implementation of VHG technology with high ethanol yield and minimised raw material losses, attention should be given to all technological stages, such as mashing, liquefaction, saccharification, and yeast fermentation. Achieving a high final ethanol concentration in the mash must be accompanied by maximum utilisation of wort sugars. This approach allows for an objective assessment of the utilisation potential of sorghum and corn under similar conditions.
Sorghum mash with a lower density may be advantageous for fermenting high-density mash (VHG). When used, it is possible to have higher ethanol yields without thickening the mash. The way the mash is treated with enzymes significantly affects the final ethanol yield. Therefore, future research should focus on developing more effective enzyme blends for sorghum, particularly when fermentation is conducted at high densities.
CONCLUSION
During fermentation under very high viscosity for 72 hours, both sorghum and corn produced ethanol at similar rates, 1.52 ± 0.05 g/(l•h) and 1.54 ± 0.02 g/(l•h), respectively. The concentrations of higher alcohols and ketones were similar for both feedstocks, whereas sorghum wort contained 15–45% less aldehydes, esters, and methanol.
During the liquefaction stage, the dynamic viscosity of sorghum wort was approximately 43% lower than that of corn. This reduction facilitated more efficient mixing and indicated the feasibility of employing higher substrate concentrations compared to corn. While this study utilized a 28% dry matter concentration, the significantly lower viscosity of sorghum suggests a higher technological threshold before reaching the critical limit of 300 mPa·s. Viscosity, enzymatic efficiency, and content of volatile by-products confirm the possibility of further increasing the sorghum wort density to increase the ethanol concentration in the fermented wort.
Furthermore, sorghum demonstrated enhanced technological efficiency by ensuring stable ethanol yields with reduced energy requirements for mash processing. Given these advantages, grain sorghum represents a viable alternative to corn for industrial bioethanol production, justifying its inclusion in renewable energy development strategies.
AUTHORS' CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: V.S.: Conceptualized the study, collected data, performed analysis, and wrote the manuscript; S.T.: study conception and design, performed analysis; T.I., Y.V., and Y.B.: Contributed to data interpretation, methodological validation, and manuscript review. All the authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| DDGS | = Distillers Dried Grains with Solubles |
| DM | = Dry Matter |
| RPM | = Revolutions per Minute |
| SHF | = Separate Hydrolysis and Fermentation |
| SSF | = Simultaneous Saccharification and Fermentation |
| VB | = Volatile By-Products |
| VHG | = Very High Gravity |
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of the article are available within the manuscript.
FUNDING
This research was supported by the National Academy of Sciences of Ukraine under the research project “Development of scientific foundations for fermentation of plant raw materials and their residues for biofuel production” (2024–2028, State Registration No. 0124U002605).
ACKNOWLEDGEMENTS
We acknowledge the suggestions of Dr. V. Kyrylenko (Institute of Food Biotechnology and Genomics, National Academy of Sciences of Ukraine, Kyiv) aimed to improve the language of the manuscript.


