All published articles of this journal are available on ScienceDirect.
The Effect of Boiled Oil (Olive Oil Produced using Boiled Olive Fruits) on the Rat's Liver
Abstract
Aims
This research aims to investigate the reduced hepatoprotective effect of virgin olive oil if olive fruits are boiled before being pressed. The hepatoprotective activity of virgin olive oil with boiled oil is also determined whether boiled oil still exhibits hepatoprotective activity in the rat’s liver.
Background
The Olive tree (Olea European L) is the most cultivated fruit tree, and olive oil is one of the main products in Jordan. The hepatoprotective activity of virgin olive oil (VOO) was earlier attributed to phenolic compounds' antioxidant activity. In some villages in Northern Jordan, villagers boil olive fruits in tap water for 10 minutes and sundry them for two weeks before oil extraction to enhance the colour and obtain an intense taste. Moreover, the locals claim that this oil has healing properties and refer to this oil as “boiled oil”.
Methods
Forty-two male Wistar albino rats were divided into six groups of 7 rats/group. Corn oil was used as a negative control. The positive control groups were fed corn oil, virgin olive oil, or “boiled oil” in addition to 3 g/Kg body weight of paracetamol (acetaminophen, N-acetyl- P -aminophenol) as a hepatotoxin on the penultimate day. The experimental groups were fed virgin olive oil (VOO) and boiled oil only. All groups were fed 7.5 ml/kg/day for 21 days. On the slaughter day, the rats were anaesthetised with ether, and blood samples were collected via heart puncture, then liver function tests, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin, were conducted. The liver was excised, washed, paper toweled and weighed to calculate the liver weight/body weight ratio (LW/BW ratio).
Results
Virgin olive oil decreased ALT significantly (p<0.05) from (108.42 ± 3.79 U/L) in the corn oil group to ((26.86 ± 8.22 U/L) in the VOO group and (71.50 ± 5.13 U/L) in the boiled group. VOO and boiled oil also affected AST similarly. AST decreased significantly from (117.07 ± 2.13 U/L) in the corn oil group to (41.16 ± 1.61 U/L) in the VOO group and (66.35 ± 4.78 U/L) in the boiled oil group. Bilirubin also decreased significantly from (0.86 ± 0.12 mg/dl) in the control group to (0.27 ± 0.02 mg/dl) in the VOO group and (0.62 ± 0.02) mg/dl) boiled oil group. Liver weight/ body weight ratio also decreased from (3.67 ± 0.10%) to (3.50 ± 0.09%) using VOO and to (3.88 ± 0.038%) using boiled oil. All positive control groups (receiving paracetamol) showed a significant increase (p<0.05) in all parameters compared with their negative controls. Moreover, VOO with paracetamol decreased ALT significantly (p<0.05) from (191.06 ± 4.23 U/L) in the corn oil group to ((70.17±17.89U/L) in the VOO group and (110.50 ± 18.69U/L) in the boiled group. AST decreased significantly from (208.94 ± 4.68U/L) in the corn oil group to (74.58 ± 3.87 U/L) in the VOO group and (116.48 ± 15.73 U/L) in the boiled oil group. Bilirubin decreased significantly from (1.65 ± 0.10 mg/dl) in the control group to (0.57±0.04 mg/dl) in the VOO group and (0.90 ± 0.08mg/dl) in the boiled oil group. LW/BW ratio also decreased from (3.76 ± 0.15%) to (3.61 ± 0.12%) using VOO and to (3.93 ± 0.12%) using boiled oil.
Conclusion
Boiling olive fruits before pressing (boiled oil) decrease hepatoprotective activity compared to VOO, but is not completely diminished.
1. INTRODUCTION
Olive oil is one of the main components of the Mediterranean diet. It is produced from olive fruit by mechanical or physical pressing [1]. The main component of olive oil is the saponifiable fraction (98-99%), composed of mono- and di-glycerides and triglycerides. The remaining is an unsaponifiable fraction (1-2%) that contains ingredients that are not used in making soaps, most of which possess antioxidant and biological activities, such as tocopherols, hydrocarbons, pigments, carotenoids, chlorophylls, sterols, triterpene alcohols, triterpenoid acids, wax esters, aliphatic alcohols, volatile and aroma compounds, and phenolic compounds are unsaponifiable fractions [2-8]. Olive oil has many beneficial health effects due to monounsaturated fatty acids, tocopherols, and polyphenols [9]. The quantity of polyphenolic compounds in olive oil is influenced by oxygen, time of pressing, processing technique, and storage conditions [10]. Moreover, high temperature and long malaxation time increase the activity of polyphenol oxidase which affects the total phenolic compounds content of the produced oil and its quality [11].
Many studies investigated the role of olive oil as a hepatoprotective agent against xenobiotic toxins and drugs. According to Amamou et al. (2015), olive oil consumption protects against cadmium-induced oxidative stress in the liver of Wister albino rats by reducing oxidative stress due to the increased activity of antioxidant enzymes [12]. Furthermore, Al-Seeni et al. (2010) reported that olive oil protects against liver damage induced by CCl4 consumption in male Wistar albino rats [13]. Also, Nakbi et al. (2010) reported that olive oil protects against oxidative stress and depletion of antioxidants caused by 2,4-dichlorophenoxyacetic acid [14]. Another study reported that Wister albino rats fed with a virgin olive oil containing diet were protected against liver damage induced by mercuric chloride [15]. Furthermore, extra virgin olive oil intake before paracetamol (acetaminophen, N-acetyl- P -aminophenol) administration in male Wistar albino rats showed reduced acetaminophen-induced toxicity in the liver by lowering the oxidative stress [16]. Overdose of paracetamol (3g/kg body weight) increases oxidative stress by being metabolised via cytochrome P450 enzymes to toxic metabolite N-acetyl- benzoquinone imine (NAPQI) that causes glutathione (GSH) depletion, which subsequently leads to alteration of hepatospecific serum markers [17]. Overdose of the acetaminophen, N-acetyl- P –aminophenol causes an imbalance between oxidants and antioxidants favoring oxidants that disrupt the redox-signaling pathway and cause overproduction of reactive oxygen species (ROS) and free radicals beyond the physiological capability to detoxify or repair [18, 19].
In some villages in Northern Jordan, locals boil olive fruits and sun-dry them for two weeks before pressing them to produce traditional olive oil with intense colour and enhanced taste, locally known as “boiled oil”. Boiled oil (BO) is generated by boiling olive fruits in half barrels, covering the fruits with tap water, and boiling using firewood for ten minutes. To remove excess water, boiled olive fruits are then spread on a clean surface and exposed to sunlight for two weeks [20-24]. The strong taste of boiled oil is an acquired taste and some Jordanians find it preferable to virgin olive oil [22]. Moreover, villagers claim that boiled oil protects against certain diseases [23]. Dried boiled olive fruits are then crushed using ancient traditional stone mills to produce a paste [25, 22]. The paste is then kept at room temperature in an open container, then it is spread by hand, on pressing mats, and finally, it is pressed using mechanical hydrostatic force to enable the separation of oil droplets [26]. Oil droplets are collected after pressing and stored in 20-litre tin containers at room temperature until use. Locally, boiled oil is about 20-30% more expensive than virgin olive oil [27].
The present study aims to investigate whether boiling olive fruits and sundry them for two weeks under the sun will affect the hepatoprotective activity of the produced oil “boiled oil” by assessing alanine aminotransferase (ALT), aspartate aminotransferase (AST) activity and total bilirubin level in the blood serum, and calculate liver weight/body weight ratio which is an indicator of hepatomegaly.
2. METHODS
2.1. Animals
Male Wistar albino rats (90-140 g) were purchased from the Animal House at Jordan University of Science and Technology in Irbid. They were divided into six groups, seven rats per group. The rats were housed in metallic cages and kept in a temperature-controlled environment (25 ± 3) °C with an alternating 12-hour light-dark cycle. Rats were acclimatised for two weeks before the beginning of the experiment. During the experimental period, rats were supplied with tap water and a standard rat chow ad libitum. All approvals for experimental animals were obtained from The Animal Care and Use Committee (ACUC) at Jordan University of Science and Technology.
2.2. Chemicals
Boiled oil and virgin olive oil (VOO) samples were purchased from a local farmer from the village of Tubneh in Northern Jordan, where the virgin olive oil was produced by the mechanical extraction process from the same crop of olive fruits used in boiled oil extraction in the traditional process. The corn oil (Shams) produced by Afla International Co was purchased from the Jordanian market. Alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin kits were purchased from Randox (UK). Diethyl ether solvent was purchased from Medex (England). Serum clot activator tubes were purchased from GongDong (China). Sodium chloride powder NaCl was purchased from REACTIFS – SDS (France), and Pa00140500 DC paracetamol (acetaminophen, N-acetyl- P -aminophenol), USP EXTRA PURE was purchased from BBC Chemicals for Lab (China). Paracetamol and acetaminophen were used interchangeably throughout this study.
2.3. Experimental Design
Forty-two rats were divided randomly into six groups; each group contained seven rats as follows:
2.3.2. Group 2
The positive control group received 7.5ml/kg/day corn oil via gavage for 20 days in addition to 3g/kg body weight acetaminophen, N-acetyl- P-aminophenol on the penultimate day [17].
2.3.3. Group 3
The negative virgin olive oil group (VOO) received 7.5ml/kg/day of virgin olive oil (VOO) via gavage for 20 days.
2.3.4. Group 4
The positive virgin olive oil group (VOO) received 7.5ml/kg/day of virgin olive oil (VOO) via gavage for 20 days in addition to 3g/kg body weight acetaminophen, N-acetyl- P-aminophenol on the penultimate day [17].
2.3.5. Group 5
The negative boiled oil group (BO) received 7.5ml/kg/day of boiled oil (BO) via gavage for 20 days.
2.3.6. Group 6
The positive boiled oil (BO) group received 7.5ml/kg/day boiled oil (BO) via gavage for 20 days in addition to 3g/kg body weight acetaminophen, N-acetyl- P-aminophenol on the penultimate day [17].
2.4. Samples Collection and Organ Weight
Rats were anaesthetised using diethyl ether. Heart puncture was used to collect blood samples. Each 5 mL of blood was transferred to a labelled serum separation clot activator single-use and a sterile tube containing micronised silica that activates clotting to separate serum. Under clean but nonsterile conditions, the rat's abdomens were opened by a midline incision, and livers were excised from the upper right of the rat’s abdomen and directly transformed into ice-cold saline solution (0.9% NaCl); livers were paper towelled then weighed. Livers were kept in labelled Petri dishes and stored at -20°C.
2.5. Liver Function Tests
The activity of liver enzymes: ALT, AST, and total bilirubin level was assessed using serum samples on the same day of serum separation by using Randox kits [28].
2.6. Alanine Aminotransferase (ALT) Test
Alanine aminotransferase was assessed using an AL 1268 kit that was stored at 2-8°C. The vial containing enzyme/coenzyme/I-oxaloglutarate was reconstituted with a vial has Tris buffer with L-alanine reagent one day before usage and was stored at 2-8°C or prepared at 15-25 °C stable for 24 hours. In 1 cm light path cuvettes and using the calibrated pipette, 1 mL of reagent and 0.1 mL of sample were mixed. The cuvette was inserted into (GENESYS 10S UV-Vis) spectrophotometer and read at 340 nm. Four different absorbance readings were recorded with a 1-minute interval; the difference between consecutive absorbance and the average absorbance difference per minute was calculated. All measurements were done against a black cuvette containing air, and the records for the first two minutes were used for calculations (Randox Laboratories Ltd, UK) kit [28].
2.7. Aspartate Aminotransferase (AST) Test
Aspartate aminotransferase was tested using an AS 1267 kit that was stored at 2-8°C. The vial containing enzyme/coenzyme/I-oxaloglutarate was reconstituted with a vial containing Tris buffer with L-aspartate reagent one day before use. The vial was stored at 2-8°C or prepared at 15-25 °C stable for 24 hours. In 1 cm light path cuvettes and using the calibrated pipette, 1 mL of reagent and 0.1 mL of sample were mixed. The cuvette was inserted into the spectrophotometer and read at 340 nm. Four different records of absorbance were recorded with a 1-minute interval in between. The difference between consecutive absorbance and the average absorbance difference per minute was recorded. All readings were done against a blank cuvette containing only air. The readings for the first two minutes were used for calculations [29].
2.8. Bilirubin Test
Bilirubin was tested using a BR 411 kit stored at 15 to 25°C. In 1 cm light path cuvettes and using the calibrated pipette, 200 μL of the sample was put with 200 μL of sulphanilic acid, one drop (50 μL) of sodium nitrite and 1000 μL of caffeine were then mixed and incubated for 10 minutes in previously set water path on reaction temperature 20-25°C. A 1000 μL of tartrate was mixed and incubated for 30 minutes at 20-25°C, a cuvette was inserted into the spectrophotometer to read at 578 nm against a sample blank that is similar in all compositions and volume but not sodium nitrite. Total bilirubin was calculated using its formulas, where ATB is the absorbance of total bilirubin [30].
Total bilirubin (µmol/l) =185*ATB (578 nm)
Total bilirubin (mg/dl) = 10.8*ATB (578nm)
2.9. Liver Weight / Body Weight Ratio
Livers were excised and transferred into ice-cold saline solution (0.9% NaCl), paper towelled, weighed, and stored at -20 °C. Liver weights were used to calculate the liver weight/ body weight ratio using the following equation:
Liver weight to body weight ratio = liver weight/ body weight *100% [31].
2.10. Statistical Analysis
The statistical analysis was conducted using the statistical package for social science (SPSS, version 23.0, 2015, Chicago, IL). The one-way analysis of variance (ANOVA) assessment was performed to test the difference between different groups, followed by mean separation using LSD Analysis. Means with different superscripts a, b, and c differ significantly at (p<0.05).
3. RESULTS
3.1. Alanine Aminotransferase (ALT) Test
Fig. (1) depicts the effect of corn oil, virgin olive oil, and boiled oil on ALT levels, with their positive controls receiving 3 g/Kg body weight of paracetamol (acetaminophen, N-acetyl- P -aminophenol) on the penultimate day. As expected, a significant (p<0.05) increase in serum ALT levels was observed in all positive control groups compared to their negative controls. A significant difference (p<0.05) between the effect of VOO and BO on ALT levels was observed, where VOO caused a decrease in ALT from (108.42 ± 3.79 U/L) in the corn oil group to (26.86 ± 8.22 U/L) and (71.50 ± 5.13) U/L) in the boiled oil group. The same pattern was observed when paracetamol was used as a hepatotoxin. ALT decreased from (191.06 ± 4.23) U/L) in the corn oil group to (70.17±17.89) U/L) in the VOO group and (110.50 ± 18.69) U/L) in the boiled oil group.
3.2. Aspartate Aminotransferase (AST) Test
Fig. (2) shows the effect of corn oil, virgin olive oil and boiled oil on AST levels, with their positive controls receiving 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol on the penultimate day. Once more, a significant (p<0.05) increase in serum AST levels was observed in all positive control groups receiving acetaminophen, N-acetyl- P-aminophenol; compared to their negative controls. Whereas, virgin olive oil (VOO) caused a significant (p<0.05) decrease in AST from (117.07 ± 2.13) U/L) in the corn oil group to (41.16 ± 1.61) U/L) in VOO and (66.35 ± 4.78 U/L) in the boiled oil (BO) group. The same pattern was observed when acetaminophen, N-acetyl- P -aminophenol was used, AST decreased from (208.94 ± 4.68) U/L) in the corn oil group to (74.58 ± 3.87) U/L) in the VOO group and (116.48 ± 15.73) U/L) in the BO group.
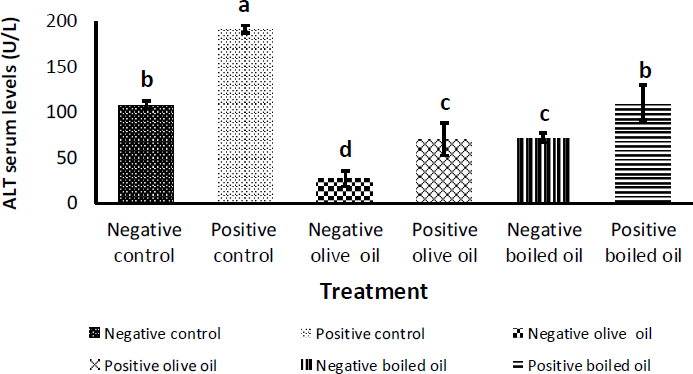
Effect of 7.5 ml/kg/day of boiled oil on ALT serum level. Positive controls of each group received 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol as a hepatotoxin on the penultimate day. Means with different superscripts a,b,c differ significantly (p<0.05). n=7.
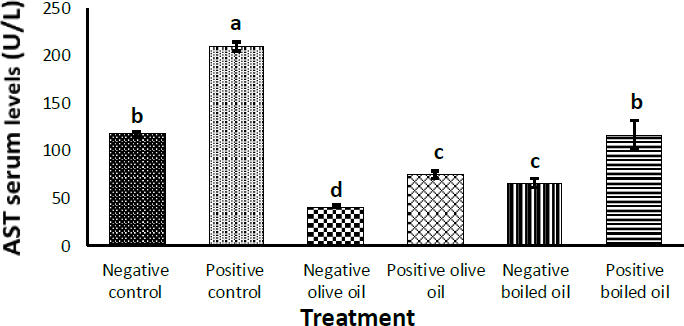
Effect of 7.5 ml/kg/day of boiled oil on AST serum levels. Positive controls of each group received 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol as a hepatotoxin on the penultimate day. Means with different superscripts a,b,c differ significantly (p>0.05). n=7.
3.3. Total Bilirubin Concentration
Fig. (3) shows the effect of corn oil, virgin olive oil and boiled oil on total bilirubin concentration, with their positive controls receiving 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol on the penultimate day. Yet again, a significant (p<0.05) increase in serum bilirubin level was observed in all positive control groups receiving acetaminophen, N-acetyl- P-aminophenol; compared to their negative controls. Virgin olive oil (VOO) caused a significant (p<0.05) decrease in bilirubin from (0.86 ± 0.12) mg/dl) in the corn oil group to (0.27 ± 0.02) mg/dl) in VOO and (0.62 ± 0.02) mg/dl) in the BO group. When acetaminophen, N-acetyl- P -aminophenol was used to induce liver damage, bilirubin decreased from (1.65 ± 0.10) mg/dl) in the corn oil group to (0.57±0.04) mg/dl) in the VOO group and (0.90 ± 0.08 mg/dl) in the boiled oil group.
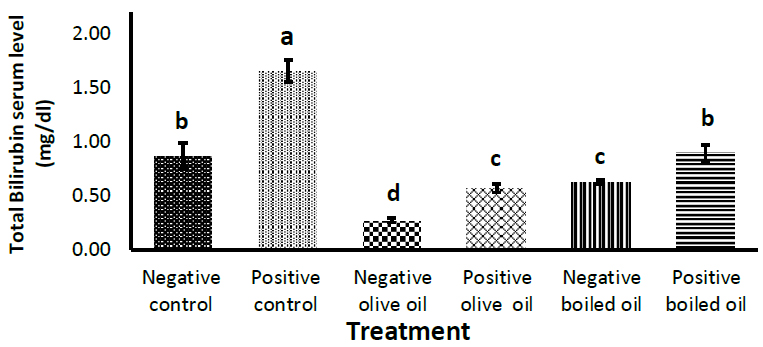
Effect of 7.5 ml/kg/day of boiled oil on total bilirubin serum levels. Positive controls of each group received 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol as a hepatotoxin on the penultimate day. Means with different superscripts a,b,c differ significantly (p>0.05). n=7.
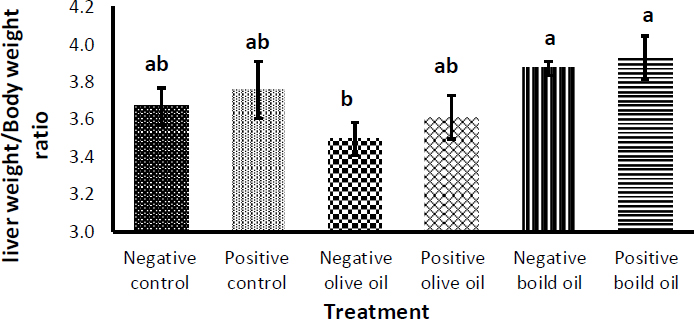
Effect of 7.5 ml/kg/day of boiled oil on the liver weight/body weight ratio. Positive controls of each group received 3 g/Kg body weight of acetaminophen, N-acetyl- P -aminophenol as a hepatotoxin on the penultimate day. Means with different superscripts a,b,c differ significantly (p>0.05). n=7.
3.4. Liver Weight/Body Weight Ratio
Fig. (4) depicts the effect of corn oil, virgin olive oil and boiled oil on liver weight/body weight ratio (LW/BW), with their positive controls receiving 3 g/Kg body weight of acetaminophen, N-acetyl-P-aminophenol on the penultimate day. There was no significant increase in LW/BW ratio in all groups receiving acetaminophen, N-acetyl- P -aminophenol compared with their negative controls because the rats were slaughtered only 24 hours after the ingestion of acetaminophen, N-acetyl- P -aminophenol and the time was not enough to cause hepatomegaly induced by acetaminophen, N-acetyl- P -aminophenol [32]. Liver weight/ body weight ratio decreased from (3.67 ± 0.10%) using corn oil to (3.50 ± 0.09%) using VOO and (3.88 ± 0.038%) using boiled oil, but the differences were not significant. The only significant difference (p<0.05) was observed between the VOO group in comparison with boiled oil, which increased from (3.50 ± 0.09%) in the VOO group to (3.88 ± 0.038%) in the boiled oil group.
4. DISCUSSION
Paracetamol (acetaminophen, N-acetyl- P –aminophenol) which is a hepatotoxin [33], was used in this study to find whether the oil extracted from boiled olive fruits (boiled oil) still possesses a hepatoprotective effect in male Wister albino rats. Hepatic enzymes (ALT, AST) and total bilirubin were estimated in serum samples of all rat groups at the end of this study. The group that received boiled oil developed a significant decrease in serum (ALT, AST and total bilirubin) in comparison with the group that received boiled oil and paracetamol, which indicates that boiled oil has a hepatoprotective effect. This activity might be attributed to different bioactive molecules present in olive fruits, such as oleic acid and phenolic compounds, which turn on several signaling pathways in hepatic cells for the prevention of oxidative stress, endoplasmic reticulum stress, mitochondrial dysfunction and inflammation resulting in resolution or prevention of hepatic injury [34, 35]. However, the hepatoprotective effect of boiled oil was significantly lower than the hepatoprotective effect of virgin olive oil. This decrease in hepatoprotective activity might be attributed to the use of high temperatures during boiling, which decreases total phenolic compound concentration and, subsequently the antioxidant activity of oil samples.
The effect of virgin olive oil on the liver in this study comes in agreement with the findings of others, that revealed that virgin olive oil decreases oxidative stress in the liver due to its high content of oleic acid, tocopherols, and polyphenols, which exhibit antioxidant properties as natural scavengers [36] Consumption of extra virgin olive oil (EVOO) was shown to improve liver enzymes due to the presence of hydroxytyrosol, a phenolic component, which prevents inflammation by inhibiting expression and activation of endothelial and intercellular adhesion molecules [37]. Oleuropein (an important component of extra virgin olive oil) inhibits cyclooxygenase-2 enzyme to bring about its anti-inflammatory action. Phenolic compounds also have anti-inflammatory activity, which causes a decrease in elevated liver enzymes [38]. These results indicate that boiled oil possesses hepatoprotective activity against oxidative stress produced by acetaminophen, N-acetyl- P –aminophenol, but it is lower than virgin olive oil, probably because boiled oil possesses significantly lower total phenolic compounds and antioxidant activity. This effect was reported in a previous study using the same specimens used in this study, and phenolic compounds decreased significantly from 8.7 mg GAE/100g in virgin olive oil to 2.47 mg GAE/100g in boiled oil. Accordingly, the antioxidant activity decreased significantly from 962 in virgin olive oil to 379 µmole/kg in boiled oil [39]. Moreover, other physical and chemical parameters of boiled oil were significantly different in comparison with virgin olive oil as follows: half-maximum inhibitory concentration (IC50) of the radical scavenging activity (RSA) was significantly higher in boiled oil samples (414 mg/ml) in comparison with VOO samples (38.9 mg/ml). Furthermore, there was a significant increase in acid value in boiled oil samples (0.943%) in comparison with VOO samples (0.518%). Likewise, the peroxide value increased significantly from 19 meq/kg in VOO in boiled oil to 500 meq/kg. Similarly, ultraviolet absorption increased significantly in boiled oil to levels considered unsuitable for human consumption [39].
Polyphenol oxidase (PPO) affects the free fatty acid (FFA) content, peroxide value (PV), and the phenol content of the virgin olive oil (VOO) [40-43]. The high temperature and long malaxation time cause a reduction of polyphenol content by the oxidative enzymatic degradation activity catalysed by polyphenol oxidases (PPO) and peroxidases (POD) and by the activity of the hydrolytic enzymes that catalyse the liberation of aglycone secoiridoids compounds from their respective glucoside forms [44-46]. Low temperatures and time, ranging between 30 and 45 mins, were the optimal operative conditions for the malaxation [40]. Whereas boiled oil production takes longer without the control of the gaseous exchanges during the operation. A negative correlation between total phenolic compounds and the time and temperature of malaxation was reported using virgin olive oil (VOO) [41-43]. Therefore, it can be speculated that the loss of hepatoprotective activity could be attributed to the loss of phenolic compounds and antioxidant activity in the boiled oil, and the claim that boiled oil is more beneficial than virgin olive oil does not apply to the hepatoprotective activity of virgin olive oil (VOO).
CONCLUSION
This study concludes that boiled oil intake before acetaminophen, N-acetyl- P –aminophenol administration in male Wister albino rats reduced acetaminophen, N-acetyl- P –aminophenol-induced toxicity in the liver by lowering ALT, AST, and total bilirubin levels and improving antioxidant defense system. This finding suggests that including boiled oil in the diet may offer benefits in terms of reducing liver damage during acetaminophen, N-acetyl- P –aminophenol toxicity, but this activity was less than the hepatoprotective activity of virgin olive oil.
AUTHORS’ CONTRIBUTION
T.A.: Data collection; H.S.: Analysis and interpretation of results; S.J.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| VOO | = Virgin olive oil |
| PPO | = Polyphenol oxidase |
| RSA | = Radical scavenging activity |
| BO | = Boiled oil |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
All experiments were performed after acquiring the approval of the Animal Care and Use Committee (ACUC) at Jordan University of Science and Technology, approval no. 12/29/4/C.CLAP, Jordan.
HUMAN AND ANIMAL RIGHTS
All the animal experimentation was performed in accordance with the GUIDE for the care and use of laboratory animals.
This study adheres to internationally accepted standards for animal research, following the 3Rs principle. The ARRIVE guidelines were employed for reporting experiments involving live animals, promoting ethical research practices.
AVAILABILITY OF DATA AND MATERIALS
The data sets used and/or analysed during this study will be available from the corresponding author [S.J] upon request.
FUNDING
The Deanship of Research/ Jordan University of Science and Technology, Jordan supported this work with Grant number 20210385.
ACKNOWLEDGEMENTS
Declared none.


