All published articles of this journal are available on ScienceDirect.
Genetic Diversity of Omani Durum Wheat (Triticum turgidum sub sp. durum) Landraces
Abstract
Introduction:
The Sultanate of Oman is rich in diversity of the most important crops like wheat, which not only has a global significance but is also regarded as one of the strategic crops in the country. The country has an ancient cultivation history of both bread wheat (Triticum aestivum L.s.l.) and durum wheat (Triticum turgidum sub sp. durum) because of its characteristic location on the eastern edge of the Arabian Peninsula. Wheat landraces constitute the prime genetic resources of cultivated wheat not only in Oman but also in several MENA (the Middle East and North Africa) countries. Indigenous landraces have paramount significance for their potential utilization in crop improvement and conservation programs. Hence, the present study was undertaken to subject 17 indigenous durum wheat accessions for analyses of diversity to select parents for hybridization in national crop improvement programs.
Materials and Methods:
The trial was conducted consecutively for two cropping seasons (2017-2018 and 2018-2019) during winter from November to March on the layouts of a loamy soil site under sprinkler irrigation system in Augmented Design with five check varieties replicated five times randomized and distributed throughout the experimental area under spacing and crop husbandry practices as per national recommendations. The data on 9 quantitative (Plant descriptors) and 6 qualitative traits on the presence (score 1) or absence (score 0) of pigmentation on 6 plant parts were collected. These traits were subjected to both Principal Component Analysis (PCA) and Hierarchical Cluster Analysis (HCA) to comprehend the contribution of these characters towards diversity and form prime diverse clusters from 17 indigenous durum wheat landraces to select appropriate parents for crossing.
Results:
The results indicated that indigenous durum wheat accessions were significantly different (p>0.05) with respect to all the quantitative characters except the number of tillers. Of 36 combinations of associations among 9 agro-morphological characters’ studied, only six correlations involving four characters viz. tiller no., spikelets/ spike, grains/spike, and grain length were found significant (p<0.05). The results of two multivariate analyses indicated the formation of four diverse clusters with different compositions of accessions, thus not supporting each other in discerning diversity. The parents were selected for hybridization for improving characters of growth for higher yield or productivity with one or two identifying markers of pigmentation on plant parts.
Conclusion:
The indigenous durum wheat landraces / accessions were found to be more diverse and potential for use in the national crop improvement programs for higher productivity.
1. INTRODUCTION
Wheat (Triticum sp.) is one of the most important food crops of the world, which is known to provide nearly 44% of the total edible dry matter and 40% of food crop energy consumed by the most developing countries of the world [1]. Of the seventeen different wheat species, only three species, namely allohexaploid Triticum aestivum, known as bread wheat, whereas other two allotetraploid species, Triticum durum (Triticum turgidum ssp. durum) and Triticum dicoccum, known internationally for pasta making and locally in specific food preparations, are cultivated in the world. Of the world wheat cultivated area, Triticum aestivum occupies 90%, whereas of the remaining two species, Triticum durum contributes 9-10% and Triticum dicoccum contributes to a negligible extent [2, 3]. As for the production at a global level, the production of wheat was estimated to be 735 million tons in 2018 to 2019, of which bread wheat accounted for about 94%. In 2017-2018, the production of durum wheat Triticum turgudum ssp. durum was recorded 37.5 million tons with more than a 5% share of the global wheat production [4-6]. Countries in the Mediterranean and Southern Europe, the Balkans, North Africa, and southwest Asia, where initial wheat domestication and cultivation are observed, the durum wheat cultivars are still predominant [7-11]. These indigenous durum wheat cultivars form an important source of genetic materials due to their ease in adapting to adverse biotic and abiotic stresses and grain quality. These could be the products of natural selection through domestication performed by the farmers through ages [4].
The Sultanate of Oman is rich in diversity of the most important crops like wheat, which not only has a global significance but is also regarded as one of the strategic crops in the country [12]. It is grown in diversified environments, as evidenced by the number of indigenous accessions collected during several collecting missions undertaken in Oman involving international organizations like the Food & Agriculture Organization (FAO) of the United Nations (UN) [13-16]. This is because of its position in the Middle East and its ancient trade relations tracing back to 3000 BCE with Iraq in the fertile crescent region that covers Mesopotamia, the home to the earliest known human civilizations and origin of several wheat species, including allohexaploid bread wheat (6x=2n=42) around 4500 BCE [17-19]. Durum wheat (Triticum turgidum sub sp. durum (4x=2n=28) having two genomes, A and B twice in its somatic cells (AABB), was domesticated at the same time [20, 21].
There exists enormous variation among different indigenous accessions of durum wheat in terms of quantitative characters that are directly related to yield and yield-related traits or qualitative traits like pigmentation and other descriptors of wheat, which are very important for crop improvement programs through breeding and selection [22-26]. Among the yield-related traits, number of tillers, number of grains/ spike, and test weight are known to influence directly and positively to the grain yield [25, 27-29]. Several approaches and analyses like those of principal components, clusters, and factors have been suggested by many researchers for estimation of genetic diversity or distances among and between groups of cultivars studied based on not only morphological and growth attributes [29-34] but also molecular markers [4, 35, 36]. Most of these authors have used Hierarchical Cluster Analysis (HCA) to estimate genetic dissimilarity and similarity, and Factor Analysis (FA)/ Principal Component Analysis (PCA) to determine the factors that contribute to the variation of quantitative characters in durum wheat. There have been no studies conducted so far on understanding the genetic diversity among the available indigenous durum landraces of Oman. Hence, in the present study, indigenous durum wheat landrace accessions repatriated from the USDA gene bank were investigated to estimate the potentiality of genetic diversity using both HCA and FA/PCA for the selection of appropriate parents for hybridization programs to improve the productivity of durum wheat.
2. MATERIALS AND METHODS
2.1. Material and Details of Conducting Experiments
Seventeen indigenous durum wheat (Triticum turgidum ssp. durum) landraces of the USDA accessions, repatriated by the Oman Animal & Plant Genetic Resources Center (OAPGRC) of the Research Council from USDA gene bank, where these accessions were deposited by international FAO collectors during their joint MAF-FAO collecting missions during the 1990s from different governorates of Oman, were studied (Table 1). The trial was conducted consecutively for two cropping seasons (2017-2018 and 2018-2019) during winter from November to March on a loamy soil site under sprinkler irrigation system at Agriculture Research Station, Jimah in Al Bahla wilayat of Al-Dakhliyah (Interior Oman) governorate. These durum wheat accessions were planted in the plots of 3-m three rows at a spacing of 0.2 m between rows and 0.15 m between plants under Augmented Design with five check varieties (WQS-302; WQS-305; WQS-308; Jimah-1 and Jimah-110) replicated five times. All the crop husbandry practices were followed according to the national recommendations of the Ministry of Agriculture & Fisheries (MAF), Oman [37], to raise a successful crop. The qualitative traits of pigmentation were recorded at the respective growth stage of the plants in each plot when the pigmentation on the plant parts was observed as intense and clear. The days to flowering and maturity were recorded when each plot attained about 50% flowering and 90% maturity of grains. The characters viz. tiller number/plant, spike density, spikes/spike, grains/spike, grain length (mm), grain width (mm), and 1000-grain weight (g) were measured based on the guidelines in Descriptors of Wheat (revised) (IBPGR) [38].
| S. No. | USDA Accession No. | Village | Wilayat | Governorate | Latitude (N) |
Longitude (E) |
Altitude (m) |
|---|---|---|---|---|---|---|---|
| 1 | PI 532239 | 2km SE of Sohar, N. Batinah Province. | Sohar | Batinah North | 24.36667 | 56.75 | 1 |
| 2 | PI 532242 | 32km SW of Majis, W. Hajar Province. | Sohar | Batinah North | 24.16667 | 56.333333 | 500 |
| 3 | PI 532279 | 20km N of Birkat al Mawz, Jebel Akhdar Province. | Nizwa | Al Dakhiliya | 23.16667 | 57.666667 | 1700 |
| 4 | PI 532281 | 20km N of Birkat al Mawz, Jebel Akhdar Province. | Nizwa | Al Dakhiliya | 23.16667 | 57.666667 | 1800 |
| 5 | PI 532287 | 50km N of Bayah, Musamdam Province. | Khasab | Musandam | 25.83333 | 56.333333 | 400 |
| 6 | PI 532288 | 10km E of Khasab, Musandam Province. | Khasab | Musandam | 26.33333 | 56.333333 | 200 |
| 7 | PI 532289 | 30km S of Khasab, Musandam Province. | Khasab | Musandam | 26.16667 | 56.166667 | 1200 |
| 8 | PI 532291 | 30km S of Khasab, Musandam Province. | Khasab | Musandam | 26.16667 | 56.166667 | 1200 |
| 9 | PI 532292 | 30km S of Khasab, Musandam Province. | Khasab | Musandam | 26.16667 | 56.166667 | 1200 |
| 10 | PI 532303 | Veterinary Research Station, Salalah, Southern Province. | Salalah | Dhofar | 17.0175 | 54.082778 | 14 |
| 11 | PI 602425 | 30km W of Rustaq, Western Hajar. | Rustaq | Batinah South | 23.33333 | 57.333333 | 700 |
| 12 | PI 532300 | 2km NE of Al Hamra, Interior Province. | Al Hamra | Al Dakhiliya | 23.16667 | 57.333333 | 500 |
| 13 | PI 532302 | Research Department, Ministry of Agriculture, Salalah, Southern Province. | Salalah | Dhofar | 17.0259 | 54.11723 | 0 |
| 14 | PI 532304 | 30km W of Rustaq, Western Hajar Province. | Rustaq | Batinah South | 23.33333 | 57.333333 | 700 |
| 15 | PI 532305 | 60km SW of Sohar, Western Hajar Province. | Yanqul | Al Dhahirah | 23.83333 | 56.333333 | 500 |
| 16 | PI 532306 | 20km N of Birkat al Mawz, Jebel Akhdar Province. | Nizwa | Al Dakhiliya | 23.16667 | 57.666667 | 1750 |
| 17 | PI 532307 | Wadi Quarayat Res. Stat., Al Kauril, Jaalan and Sur Province. | Jalan Bani Bu Hassan | Alsharqya South | 22.16667 | 59.166667 | 200 |
2.2. Statistical Analyses
Analysis of variance, correlation analysis, HCA through agglomerative hierarchical clustering (AHC) and FA/ PCA were performed by applying XLSTAT –software for Excel [39]. Factor analysis aimed for the estimation of commonality from the quantum of variance by the highest correlation coefficient in each array [40] and adopted Euclidian distance as a measure of dissimilarity and the unweighted pair-group average method as the clustering algorithm, whereas PCA estimated the number of factors. The software adapted varimax or orthogonal rotation to define each factor as a distinct cluster of correlated variables and determined factor loadings of the rotated matrix, percent of variability contributed by each factor, and commonalities of each variable. In the present study, a combined analysis of variance of two-year data of check varieties indicated an insignificant effect of GxE (genotype and environment/year) interaction (p>0.05) for each quantitative character. PCA was performed on the correlation matrix between two-year means of agro-morphological characters and scores of presence (1) and absence (0) of anthocyanin pigmentation on six plant parts in indigenous durum accessions. The data on plant height, growth habit, and awns were not included in statistical analysis as all the accessions were tall, erect, and awned, respectively, in their morphological features. Similarly, grain color was not included in the analysis because of the absence of appropriate measurement for distinctness, although landraces had variation in grain color. Landraces of wheat are all tall as they were constantly selected for a long time by the local farmers for straw traditionally to feed their livestock (Table 2).
| Accession USDA No. | Plant height* |
Days to flower |
Days to mature |
Spike- lets/ spike |
Grains/ spike |
Grain Length (mm) |
Grain width (mm) |
1000 grain weight (g) | Growth Habit* | Tiller No. | Awns* |
Spike density |
Grain Color |
Coleoptile color |
Nodal Color | Internode Color | Leaf Color | Leaf Sheath Color | Auricle |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PI 532239 | Tall | 72 | 112 | 69 | 66 | 6.6 | 3.58 | 25.5 | 3 | 4 | 7 | 7 | M.Brown | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532242 | Tall | 74 | 112 | 64 | 59 | 7.5 | 3.31 | 33.5 | 3 | 8 | 7 | 6 | M. Brown | 0 | 0 | 0 | 1 | 0 | 0 |
| PI 532279 | Tall | 68 | 115 | 92 | 87 | 7.1 | 2.80 | 37.1 | 3 | 4 | 7 | 7 | Yellow | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532281 | Tall | 70 | 118 | 87 | 85 | 5.4 | 2.40 | 35.7 | 3 | 5 | 7 | 7 | L. Brown | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532287 | Tall | 70 | 112 | 60 | 57 | 7.7 | 2.40 | 40.3 | 3 | 7 | 7 | 7 | L. Brown | 1 | 0 | 0 | 0 | 0 | 0 |
| PI 532288 | Tall | 72 | 125 | 55 | 51 | 7.7 | 3.31 | 40.3 | 3 | 6 | 7 | 7 | L. Brown | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532289 | Tall | 66 | 115 | 71 | 68 | 7.41 | 3.17 | 39.5 | 3 | 4 | 7 | 7 | Yellow | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532291 | Tall | 79 | 116 | 71 | 68 | 7.3 | 2.97 | 39.2 | 3 | 4 | 7 | 7 | Yellow | 0 | 0 | 0 | 1 | 0 | 0 |
| PI 532292 | Tall | 68 | 118 | 53 | 48 | 6.3 | 2.10 | 42.6 | 3 | 4 | 7 | 7 | L. Brown | 0 | 0 | 0 | 1 | 0 | 0 |
| PI 532303 | Tall | 74 | 112 | 36 | 33 | 9.8 | 2.34 | 32.9 | 3 | 9 | 7 | 7 | L. Brown | 1 | 0 | 1 | 0 | 1 | 0 |
| PI 602425 | Tall | 86 | 120 | 43 | 38 | 6.4 | 2.02 | 37.8 | 3 | 3 | 7 | 7 | D. Brown | 1 | 0 | 0 | 1 | 0 | 0 |
| PI 532300 | Tall | 72 | 127 | 64 | 60 | 6.5 | 2.25 | 42.6 | 3 | 4 | 7 | 7 | L. Brown | 1 | 0 | 0 | 1 | 1 | 0 |
| PI 532302 | Tall | 76 | 120 | 32 | 29 | 7.8 | 3.80 | 44.7 | 3 | 7 | 7 | 7 | L. Brown | 1 | 0 | 0 | 1 | 1 | 0 |
| PI 532304 | Tall | 74 | 115 | 46 | 44 | 8.8 | 2.43 | 30.9 | 3 | 8 | 7 | 7 | L. Brown | 1 | 1 | 1 | 1 | 1 | 0 |
| PI 532305 | Tall | 76 | 115 | 45 | 43 | 9.2 | 2.72 | 37.9 | 3 | 9 | 7 | 7 | L. Brown | 1 | 0 | 0 | 1 | 1 | 1 |
| PI 532306 | Tall | 95 | 127 | 45 | 42 | 8.2 | 2.17 | 35.4 | 3 | 8 | 7 | 7 | L. Brown | 0 | 0 | 0 | 1 | 1 | 0 |
| PI 532307 | Tall | 85 | 120 | 56 | 54 | 8.9 | 2.45 | 30.0 | 3 | 8 | 7 | 7 | L. Brown | 0 | 0 | 0 | 1 | 1 | 0 |
| - | - | - | - | - | - | - | - | 3-upright 7-Prostrate |
- | 0-awn less 3-awnletted 7-awned |
1-Very lax 2-Lax 5- Intermediate 7- Dense 9- Very dense |
L-light M-Medium D-Dark |
1-Purple 0- Green |
1- Purple 0- Green |
1- Purple 0- Green |
1- Cement color 0- Green |
1- Purple 0- Green |
1- Purple 0- Green |
3. RESULTS
3.1. Variation in Aagro-morphological and Qualitative Traits among Oman Landraces
The aggregate mean values over two cropping seasons for all agro-morphological characteristics of Oman landraces with USDA accession nos. are presented in Table 2. The accessions were found to be significantly different (p<0.05) in respect of all the quantitative characters except the number of tillers, and had a higher degree of variation in respect of most of the studied quantitative characters (Table 3). These accessions were similar with respect to plant stature (tall), erect growth habit, and presence of awns (Table 2). The accessions were the most diverse in respect of spikelets/ spike and grains/spike, respectively, from 32 and 29 (PI 532302) to 92 and 87 (PI 532279) in comparison with other characters studied.
There existed variation also for seed characteristics and seed dimensions among the durum accessions, studied especially with respect to test weight and seed length besides grain color. The accession, PI 532239 showed the lowest 1000-grain weight with 25.5 g, whereas the highest 1000-grain weight was found in the accession PI 532302 (44.7 g), followed by two accessions, PI 532292 and PI 532300 with 42.6 g and another two accessions PI 532287 and PI 532288 with 40.3 g. The grain length, however, varied from 5.4 mm (PI 532281) to 9.8 mm (PI 532303), whereas grain width varied from the lowest of 2.02 mm as in the case of PI 602425 to the highest of 3.80 mm for PI 532302 (Tables 1 and 2). In respect of grain color, 11 accessions had light brown grain color followed by three accessions with yellow, 2 accessions were medium brown, and 1 accession with dark brown in their grain color (Table 2).
Generally, it was observed that these indigenous landraces had the extreme value for a character or few expressing in opposite trend for other traits. For example, PI 532302, with the least number of spikelets or grains per spike, had higher values of grain dimensions and other characters. There was a high diversity among durum landraces in seed color, seed shape, and seed size.
In respect of qualitative traits, as many as 12 accessions have coleoptile purple followed by 7 accessions having leaf sheath purple, 2 accessions with internode purple, and one accession, each with node and auricle pigmentation (Table 2).
3.2. Correlation among Agro-morphological Traits
Phenotypic characters played a significant role during the introduction of crop plants as traits such as plant height, days to flowering and maturity, spike, and seed characters were selection criteria during domestication. In this respect, an overview based on the association between characters could have a strong connection with the introduction of the crop species. Statistical comparisons of relationships among nine agronomical and morphological traits for 17 indigenous durum landraces are shown in Table 4.
Of 36 combinations of associations among 9 agro-morphological characters studied, only six correlations involving four characters viz. tiller no., spikelets/ spike, grains/spike, and grain length, were found significant (p<0.05) (Table 4). The correlations of tiller no. with spikelets /spike (-0.537 *) and grains/ spike (-0.508*) were negative and significant, whereas those with grain length was positive (+0.833**) and highly significant. Similarly, grains/spike was positively, highly, and significantly correlated with spikelets/spike (+0.998**), whereas the associations of grain length with spikelets/spike (-0.581*) and grains/spike (-0.556*) were negative and significant (p<0.05).
|
Sl. No. |
Characters | Minimum | Maximum | Mean | F-Test |
Std. Error. (SE) (16 d.f.) |
LSD (p=0.05) |
CV (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | Days to flowering | 66 | 95 | 75.12 | * | 7.49 | 18.53 | 6.37 |
| 2 | Days to maturity | 112 | 127 | 117.59 | * | 5.03 | 12.44 | 4.47 |
| 3 | Tiller No. | 3 | 9 | 6.00 | NS | 2.09 | - | 34.87 |
| 4 | Spike density | 6 | 7 | 6.94 | * | 0.24 | 0.59 | 3.50 |
| 5 | Spikelets/spike | 32 | 92 | 58.18 | * | 16.65 | 41.19 | 28.62 |
| 6 | Grains/spike | 29 | 87 | 54.82 | * | 16.54 | 40.92 | 30.16 |
| 7 | Grain length (mm) | 5.4 | 9.8 | 7.57 | * | 1.16 | 2.86 | 15.30 |
| 8 | Grain width (mm) | 2.02 | 7.80 | 2.72 | * | 1.33 | 3.29 | 2.09 |
| 9 | 1000 grain weight (g) | 25.5 | 44.7 | 36.82 | * | 5.05 | 12.49 | 13.71 |
| Variables/ Characters |
Days to maturity | Spikelets/ spike |
Grains/ spike |
Grain Length (mm) | Grain width (mm) | 1000 grain weight (g) | Tiller No. |
Spike density |
|---|---|---|---|---|---|---|---|---|
| Days to flowering | 0.460ns | -0.462 ns | -0.455 ns | 0.283 ns | -0.098 ns | -0.209 ns | 0.295 ns | 0.038 ns |
| Days to maturity | 1 | -0.181 ns | -0.189 ns | -0.188 ns | -0.008 ns | 0.392 ns | -0.137 ns | 0.287 ns |
| Spikelets/spike | - | 1 | 0.998** | -0.581* | -0.251 ns | -0.107 ns | -0.537* | -0.090 ns |
| Grains/spike | - | - | 1 | -0.556* | -0.248 ns | -0.130 ns | -0.508* | -0.065 ns |
| Grain Length (mm) | - | - | - | 1 | 0.044 ns | -0.261 ns | 0.833** | 0.004 ns |
| Grain width (mm) | - | - | - | - | 1 | 0.281 ns | 0.087 ns | -0.069 ns |
| 1000 grain weight (g) | - | - | - | - | - | 1 | -0.267 ns | 0.169 ns |
| Tiller No. | - | - | - | - | - | - | 1 | -0.246 ns |
3.3. Multivariate Analyses
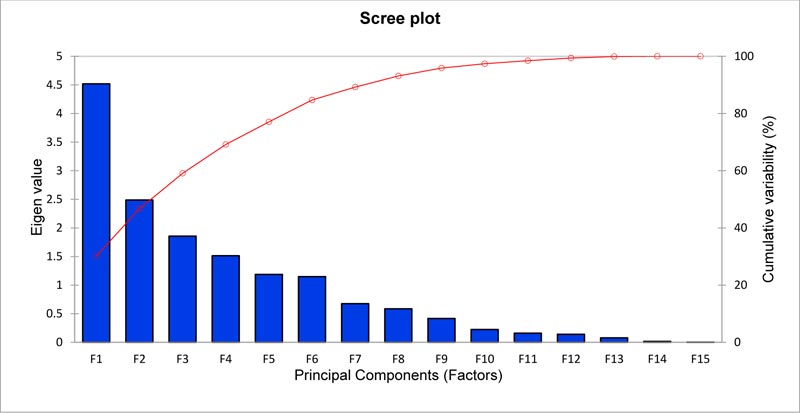
Multivariate analyses have been applied to measure the diversity in durum wheat germplasm accessions and evaluate the relative contributions of different characters to the total variability in a crop germplasm collection. These analyses enable germplasm accessions to be classified into groups/ clusters with similar characters. In this study, the principal component analysis (PCA) based on 9 agro-morphological traits and presence or absence of pigmentation on six plant parts was used to discern the patterns of variation within 17 durum wheat accessions collected from different governorates of Oman during the early 1990s. Using a PCA based on the correlation matrix, it appeared that the first eight principal components accounted for 93.122% of the total variance (Table 5) as depicted in the scree plot (Fig. 1).
The first principal component (PC1) accounted for approximately 30.127% of the total phenotypic variation (Table 5) which was influenced to the extent of 76.839% by four quantitative characters and one qualitative character viz. grain length (16.943%), spikelets/spike (15.455%), tiller no. (15.271%), grains/spike (14.598%), and leaf sheath color (14.572%) (Table 6). PC2 accounted for 16.579% (Table 5) phenotypic variability, which was contributed mainly by days to maturity (26.565%), days to flowering (13.348%), and internode color (13.086%) to the extent of 52.999% (Table 6). However, PC3, which accounted for 12.370%, was built from coleoptile pigmentation (33.057%), spike density (19.197%), 1000-grain weight (17.601%), and days to flowering (11.594%), contributing in total 81.449% (Table 6).
| PC1 | PC2 | PC3 | PC4 | PC5 | PC6 | PC7 | PC8 | |
|---|---|---|---|---|---|---|---|---|
| Days to flowering | 0.205 | 0.365 | -0.340 | 0.180 | 0.032 | -0.173 | -0.291 | -0.227 |
| Days to maturity | -0.003 | 0.515 | 0.053 | 0.340 | 0.010 | -0.048 | 0.102 | 0.337 |
| Spikelets /spike | -0.393 | -0.208 | -0.128 | 0.067 | 0.204 | 0.048 | -0.103 | 0.389 |
| Grains /spike | -0.382 | -0.216 | -0.126 | 0.076 | 0.228 | 0.056 | -0.132 | 0.409 |
| Grain Length (mm) | 0.412 | -0.106 | -0.063 | -0.127 | 0.136 | -0.034 | -0.076 | 0.215 |
| Grain width (mm) | 0.042 | 0.169 | 0.271 | -0.345 | -0.342 | 0.499 | -0.472 | 0.108 |
| 1000 grain weight (g) | -0.098 | 0.316 | 0.420 | -0.200 | -0.114 | -0.090 | 0.566 | 0.246 |
| Tiller No. | 0.391 | -0.075 | -0.165 | -0.239 | 0.100 | 0.040 | -0.004 | 0.355 |
| Spike density | 0.028 | 0.086 | 0.438 | 0.423 | 0.264 | -0.251 | -0.305 | -0.034 |
| Coleoptile color | 0.001 | -0.218 | 0.575 | 0.078 | 0.150 | 0.120 | -0.218 | -0.117 |
| Nodal Color | 0.191 | -0.252 | -0.004 | 0.447 | -0.109 | 0.434 | 0.332 | -0.046 |
| Internode Color | 0.307 | -0.362 | 0.066 | 0.275 | -0.189 | 0.010 | 0.137 | 0.041 |
| Leaf Color | -0.162 | 0.302 | -0.180 | 0.207 | 0.165 | 0.630 | 0.054 | -0.138 |
| Leaf Sheath Color | 0.382 | 0.153 | 0.049 | 0.140 | 0.158 | 0.115 | -0.108 | 0.417 |
| Auricle | 0.143 | 0.041 | 0.084 | -0.293 | 0.745 | 0.161 | 0.210 | -0.244 |
| Eigen value | 4.519 | 2.487 | 1.855 | 1.513 | 1.186 | 1.147 | 0.675 | 0.586 |
| Variability (%) | 30.127 | 16.579 | 12.370 | 10.088 | 7.905 | 7.648 | 4.502 | 3.904 |
| Cumulative % | 30.127 | 46.705 | 59.075 | 69.163 | 77.068 | 84.715 | 89.218 | 93.122 |
| - | PC1 | PC2 | PC3 | PC4 | PC5 | PC6 | PC7 | PC8 |
|---|---|---|---|---|---|---|---|---|
| Days to flowering | 4.205 | 13.348 | 11.594 | 3.253 | 0.101 | 3.000 | 8.452 | 5.162 |
| Days to maturity | 0.001 | 26.565 | 0.277 | 11.581 | 0.009 | 0.232 | 1.037 | 11.379 |
| Spikelets/spike | 15.455 | 4.326 | 1.636 | 0.443 | 4.147 | 0.229 | 1.053 | 15.102 |
| Grains/spike | 14.598 | 4.679 | 1.578 | 0.573 | 5.203 | 0.313 | 1.731 | 16.767 |
| Grain Length (mm) | 16.943 | 1.120 | 0.393 | 1.625 | 1.853 | 0.115 | 0.576 | 4.629 |
| Grain width (mm) | 0.174 | 2.856 | 7.339 | 11.911 | 11.686 | 24.907 | 22.290 | 1.174 |
| 1000 grain weight (g) | 0.954 | 9.978 | 17.601 | 4.008 | 1.290 | 0.811 | 32.041 | 6.076 |
| Tiller No. | 15.271 | 0.558 | 2.730 | 5.715 | 1.007 | 0.159 | 0.001 | 12.590 |
| Spike density | 0.076 | 0.743 | 19.197 | 17.896 | 6.993 | 6.293 | 9.325 | 0.117 |
| Coleoptile color | 0.000 | 4.773 | 33.057 | 0.610 | 2.236 | 1.436 | 4.763 | 1.362 |
| Nodal Color | 3.652 | 6.350 | 0.002 | 19.975 | 1.187 | 18.831 | 11.021 | 0.212 |
| Internal Color | 9.436 | 13.086 | 0.429 | 7.578 | 3.583 | 0.010 | 1.865 | 0.165 |
| Leaf Color | 2.611 | 9.093 | 3.226 | 4.296 | 2.725 | 39.749 | 0.288 | 1.911 |
| Leaf Sheath Color | 14.572 | 2.355 | 0.237 | 1.959 | 2.481 | 1.329 | 1.158 | 17.378 |
| Auricle | 2.052 | 0.170 | 0.703 | 8.578 | 55.500 | 2.585 | 4.398 | 5.977 |
The first two PCAs that contributed 46.705% of the total variation present in the accessions were used to draw a biplot graph to understand the pattern of distribution of accessions in the four quadrants of the graph. Thus, PCA discriminates all the Oman durum wheat landraces into four clusters scattered respectively over four quadrants of the biplot graph (Fig. 2). The graph also represents eigenvectors of the characters that greatly influence each axis of the PC1 and PC2 in terms of length that is proportional to its magnitude of influence on that axis [29, 41, 42]. Accordingly, quadrant I (+,+) consisting of four accessions viz. PI 532302, PI 532305, PI 532306, and PI 532307 formed cluster I, which was highly influenced by days to flowering and leaf sheath color. The quadrant II (-,+) consisting of five accessions viz. PI 532288, PI 532291, PI 532292, PI 532300, and PI 602425 formed cluster II, which was highly influenced by days to maturity and leaf color. The quadrant III (-,-) consisting of six accessions viz. PI 532239, PI 532242, PI 532279, PI 532281, PI 532287, and PI 532289 formed cluster II, which was highly influenced by spikelets/spike and grains/spike whereas cluster IV was formed of only two accessions located in quadrant IV (+,-) which were greatly influenced by internode color and grain length.

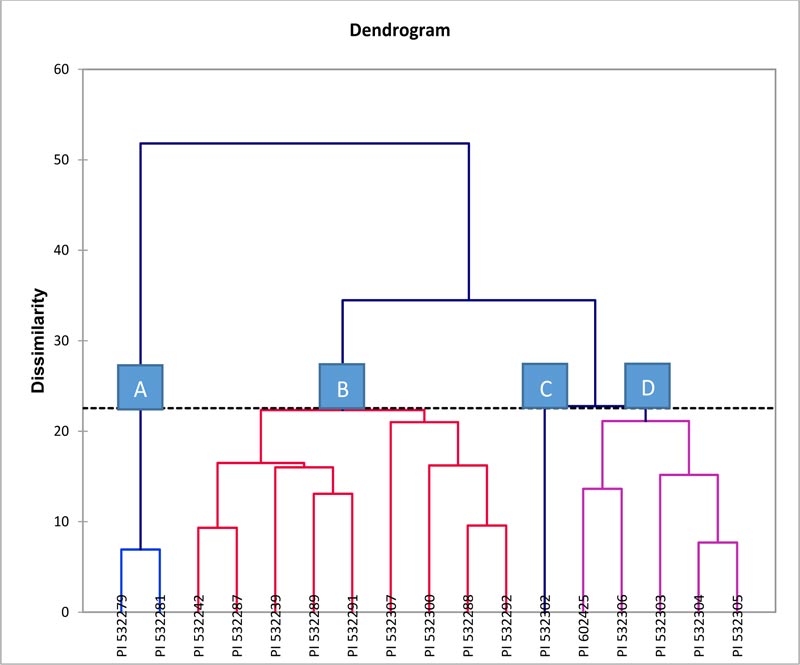
The cluster analysis has been applied to explain the relationship among the indigenous durum wheat landraces, as different accessions within a cluster were presumed to be more closely related to each other in terms of the characters under investigation than those accessions of other clusters. The nature of association among 17 indigenous durum wheat landraces signifying diversity was revealed by Wards’ method of cluster analyses on the basis of Euclidian distance coefficients (Fig. 3) for 9 agro-morphological and 6 pigmentation traits (Table 5). As against the classificatory pattern indicated by the PCA, the HCA of Wards’ method showed the different composition of four classes (clusters) of indigenous durum wheat accessions as A, B, C, and D. (Fig. 3).
Cluster A contained only two landraces (PI 532279 and PI 532281); cluster B consisted highest of nine accessions, viz. PI 532242, PI 532287, PI 532239, PI 532289, PI 532291, PI 532307, PI 532300, PI 532288, and PI 532292. Cluster C was formed of only one accession, PI 532302, whereas cluster D consisted of five accessions viz. PI 602425, PI 532306, PI 532303, PI 532304, and PI 532305.
In both the pattern of clustering, it was observed that the landraces collected from different governorates of Oman were distributed over all clusters, indicating the existence of more diversity.
4. DISCUSSION
The Sultanate of Oman has an ancient cultivation history of both bread wheat (Triticum aestivum L.s.l.) and durum wheat (Triticum turgidum ssp. durum) because of its characteristic location on the eastern edge of the Arabian Peninsula [13, 43-45]. Wheat landraces constitute the prime genetic resources of cultivated wheat not only in Oman but also in several MENA (the Middle East and North Africa) [42, 46-52]. Indigenous landraces have paramount significance for their potential utilization in crop improvement and conservation programs. A large number of wheat landraces have been collected from different parts of Oman during collecting missions during the 1980s and 1990s after Oman’s inclusion in worldwide activities for the collection of plant genetic resources under the FAO program [13]. The T. turgidum USDA accessions used in the present investigation were part of the collected genetic material during the 1990s [13]. Although the wealth of the Omani landraces has been emphasized [53], the morphological variation, tolerance to biotic and abiotic stresses, and their quality characters have not been significantly exploited in crops like wheat [52, 54]. Besides, information on the extent and pattern of genetic diversity in these durum wheat accessions is not available.
The results of the present investigations clearly revealed that 17 indigenous accessions differed significantly from one another with respect to all morpho-agronomic and pigmentation characters and showed a considerable level of phenotypic variation in terms of diversity (Tables 2-6; Figs. 1-3). In view of the requirement to use new traits in breeding procedures and statistical methods for integration of disciplinary investigation [49], in the present study, the pigmentation on different parts of the plant as presence (score 1) or absence (score 0) has been considered in the analyses of diversity [55] for their potential use as genetic identification markers in DUS tests of varieties [56-58]. Our results are in agreement with those of previous studies underlining the existence of genetic variability or diversity among durum wheat accessions studied not only in terms of agro-morphological characters like tiller number, spikelets/spike, grains/spike, 1000 grain weight [3, 25, 29, 59-62] but also for days to flowering and maturity [3, 25].
Correlation coefficients for the agro-morphological and qualitative characters were examined for potential correlations to determine whether the selection for stability in one character might influence the stability in other characters of growth of the crop. In this study, only two of the six significant correlations were found positive among the different agro-morphological and quality traits in durum wheat indigenous accessions investigated (Table 4). Accordingly, a positive association among the characters indicated that improvement of one character might simultaneously improve the other desired trait [29, 63]. For example, a positive correlation among the traits related to inflorescence, such as spikelets/spike and grains/spike showed that an improvement in physical properties of the inflorescence also results in an improvement in the number of grains that contribute to yield. Conversely, negative associations between the growth and yield attributes would help in maintaining the magnitude of the characters in balance for achieving optimum yield. For instance, spikelets/ spike or grains/ spike could be balanced to achieve the required grain length as correlations between spikelets/grains and grain length are negative and significant. Similarly, the tiller number should be optimum to get maximum spikelets and grains (Table 4). Similar observations were done by other researchers in their durum wheat germplasm [64].
The structure of the genetic diversity among the set of 17 indigenous durum wheat accessions representing six governorates of Oman extending from north of the country to its south, was assessed by multivariate analyses such as PCA and CA based on aggregate phenotypic means over two cropping seasons. Using a PCA based on the correlation matrix, it appeared that the first eight principal components accounted for over 90% of the total variance of which the first two components contributed 46.705%, which was considered to be substantial to scatter the positions of accessions over the four quadrants of the biplot graph (Fig. 2). Such a magnitude of the contribution of each of the first three PCs rightly agreed with the results of research conducted in 2014 [65], which indicated the contribution of the PC1, PC2, and PC3 as 25.9%, 17.1%, and 13.3%, respectively and that in 2018 [42] which found PC1, PC2 and PC3 contributing 45.13%, 17.85%, and 14.71%, respectively in wheat germplasm studied. Similar results were also obtained by other researchers [66-71]. In PC1, tiller number, grain length, spikelets/spike, grains/spike, and leaf sheath color were effective main sources of variation, whereas, in PC2, accessions were discriminated mainly through days to flowering, days to maturity and internode pigmentation. This is because, in PCA, differentiation of accessions into different clusters was ensured with a relatively high contribution of few characters rather than a small contribution from each character [72, 73].
In the present investigation, the results of two multivariate analyses viz. PCA and CA based on Euclidian distances showed different patterns of clustering, and they did not appear to support each other. PCA analysis formed four clusters viz. cluster I of 4 accessions, cluster II of 5 accessions, cluster III of 6 accessions, and cluster IV of 2 accessions (Fig. 2) while a dendrogram of cluster analysis (Fig. 3) revealed the formation of four clusters viz. cluster A of 2 accessions, cluster B of 9 accessions, cluster C of only one accession and cluster D of 5 accessions based on the principle of discrimination each one adapted. Further, these results revealed that accessions within each cluster might belong to the same or different governorates (Figs. 2 and 3), thus suggesting the fact that there existed no clear relationship between accessions and geographical diversity. For instance, in the composition of clusters from PCA, the accessions of each of all the four clusters were found to be from different governorates. Cluster IV consisted of two accessions, namely PI 532303 and PI 532304, which belonged to Al Batinah South (Rustaq) and Dhofar (Salalah) governorates, which are about 1000 km distant. Similarly, all the four accessions of cluster I were the ones collected from the four different governorates viz. PI 532 302 (Dhofar), PI 532305 (Al-Dhahirah), PI 532306 (Al-Dakhiliya), and PI 532307 (Alsharqiya South). A similar trend of clustering was observed in cluster II and cluster-III. This might be due to the exchange of landraces between the farmers of different geographic regions either voluntarily among relatives or through trading in the markets. Conversely, the dendrogram of CA, tended to include accessions of the same geographical area or governorate in one cluster. For instance, Cluster A consisted of two accessions (PI 532279 and 532281) that belonged to Al-Dakhiliya governorate, and all the five accessions collected from Khasab such as PI 532287, PI 532288, PI 532289, PI 532291, and PI 532292 were included in Cluster-B, although these emerged from three different branches or nodes of the dendrogram. This further showed that the diversity in indigenous durum wheat accessions collected from various geographical parts of Oman might be similar or different in genetic constitution. Therefore, more emphasis on the selection has to be directed to landraces per se rather than considering geographical location, alone, as a source of diversity in this germplasm.
The results of multivariate cluster analyses revealed the existence of substantial variation among the durum wheat landrace accessions in the present study, which could be implied in the selection of appropriate accessions based on characters to be improved, as parents for producing transgressive segregants in a minimum period of time in the crop improvement program [42, 74, 75]. Accordingly, the accession PI 532306 of Al Dakhiliya can be chosen from the cluster-I as one of the parents for manipulating days to flowering with leaf sheath as identification marker; Similarly, PI 532291 and/ or PI 532 292 of Musandam can be chosen from cluster-II for improving grain weight with leaf color as marker-trait, PI 532239 of North Al- Batinah and/ or PI 532281 of Al Dakhiliya from cluster-III for improving spikelets/spike or grains/spike with coleoptile color as marker-trait, and PI 532303 or PI 532304 of Al-Dhofar governorate from cluster-IV for improving tiller number and grain length along with internode color as marker-trait. The durum wheat accessions of the present study are part of landraces of Oman, which are well known for their desirable features of bread-making quality for the local culinary items besides their resilience nature to survive abiotic stresses like drought, heat, salinity [34, 45, 47, 76]. It is interesting to note that as many as six accessions, PI 532239, PI 532242, PI 532288, PI 532289, PI 532292, and PI 532302, have been included in the core collections of USDA gene bank based on their potential characters of growth and tolerance to several diseases of wheat like stripe rusts [62]. Hence, it is essential to conserve and use these landraces for improving the production of quality wheat used in local preparations in Oman.
CONCLUSION
Seventeen indigenous durum wheat landraces/ accessions were significantly different (p<0.05) with respect to nine quantitative characters except in respect of a number of tillers and had a higher degree of variation in respect of most of these quantitative characters. Only six correlations involving four characters viz. tiller no., spikelets/ spike, grains/spike, and grain length were significant (p<0.05). Two multivariate analyses were found not supporting each other in discerning diversity, with each forming four diverse clusters with different compositions of accessions. However, their overall results led to the selection of parents for hybridization to improve the characters of growth for higher yield or productivity with one or two identifying markers of pigmentation on plant parts.
LIST OF ABBREVIATIONS
| AHC | = Agglomerative Hierarchical Clustering |
| BI | = Bioversity International |
| CA | = Cluster Analysis |
| FAO | = Food and Agriculture Organization of the United Nations |
| HCA | = Hierarchical Cluster Analysis |
| IBPGR | = International Besrueu of Plant Genetic Resources (Now, BI) |
| MAF | = Ministry of Agriculture & Fisheries, Oman |
| MENA | = Middle East and North Africa |
| OAPGRC | = Oman Animal & Plant Genetic Resources Canter, Oman |
| TRC | = The Research Council of Oman |
| PCA | = Principal Component Analysis |
| USDA | = United States Department of Agriculture |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the finding is available in Genetic Resources Information Platform (GRIP) and annual reports of Oman Animal and Plant Genetic Resources Center www.oapgrc.gov.om.
FUNDING
The authors gratefully acknowledge OAPGRC, TRC of Oman, for the financial support of the research.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors thank the directors and administrators of the Oman Animal & Plant Genetic Resources Center of The Research Council and the Ministry of Agriculture & Fisheries of Oman for their encouragement during the research.


