All published articles of this journal are available on ScienceDirect.
Inhibition of Verticillium Dahliae and Rhizoctonia Solani Growth by Solid Phosphate Sludge Amendments
Abstract
Background:
In Morocco, phosphate processing generates large quantities of sludge which amass, form fillings, reduce arable land and distort the landscape. Phosphate wash sludge is rich in mineral elements. In order to recover some of these mineral elements including phosphorus, we considered using this sludge in a nursery as a substrate.
Objective:
The aim of the study was to check the effect of solid phosphate sludge amendment on some of the main soil-borne pathogens: Verticillium dahliae Kleb. and Rhizoctonia solani Kühn.
Materials and Methods:
The pathogens have been isolated and identified in the laboratory of Plant Pathology and Postharvest Quality of INRA-Kenitra. Phosphate wash sludge used was originated from the Khouribga phosphate treatment sludge disposal site. Different concentrations of phosphate sludge (0, 10, 30, 50, and 100 g/l) were tested on a PDA-based medium, in three replicates. The inhibitory effects on mycelial growth were measured and compared with those on the soil used by nurseries (Maamora forest soil). Petri dishes with PDA alone were used as control. The same concentrations (10, 30, 50 and 100 g/kg) were tested in vivo on two varieties of tomato and on Faba Faba bean for Verticillium dahliae and Rhizoctonia solani, respectively.
Results:
The results of this study showed that there was significant growth inhibition at different concentrations of solid Phosphate sludge amendments in vitro. The highest inhibition percentage was observed at 100 g/l of phosphate sludge amendments with 42% and 75% for Verticillium dahliae and Rhizoctonia solani. The high concentrations of phosphate sludge have shown lower disease severity of Verticillium dahliae on the tomato plants and a disease index of Rhizoctonia solani on the Faba bean plants.
Conclusion:
Based on these results, we can recommend that solid phosphate sludge can be used or mixed with other substrates in nurseries.
1. INTRODUCTION
Solid Phosphate sludge refers to the product resulting from the exploitation and subsequent metallurgical treatment of phosphorus-containing ores.
This processing generates large quantities of sludge which amass, form fillings, reduce arable land and distort the landscape. The phosphate sludge contains phosphorus, minerals elements and some of the original impurities. To recover some of these mineral elements including phosphorus, we considered using this sludge in a nursery as a substrate. This substrate can increase phosphate availability and phosphate uptake by plants. This improvement in plant nutrition can enhance plant growth and, therefore, make it more resistant to disease [1].
The use of soil amendments for disease suppression is now an approach for the biological control of soil-borne plant pathogens [2-4]. Recently, the use of soil amendments to control vascular pathogens is of particular interest. Rhizoctonia solani and Verticillium dahliae pathogens are among the most devastating soil-borne pathogens, which cause many diseases in several crops of agronomic importance in the country and the world. Their symptoms depend on the virulence of the pathogen, the host, and the environmental conditions. Symptoms include epinasty, chlorosis, partial or complete wilting, stunting, and ultimately death [5, 6]. The fungal pathogens can infect a broad number of plants, as well as weeds and volunteer plants [7]. Pesticides have been used to control different soil-borne pathogenic fungi including Rhizoctonia spp. and Verticillium spp [8-10]. These chemicals can be toxic to the environment and human health [11] and many countries have restricted their usage [11, 12]. Additionally, both pathogens produce persistent structures that can survive in the soil for several years to a decade. Verticillium dahliae acts as microsclerotia and Rhizoctonia solani as a macrosclerotia in the absence of a susceptible host. When the environmental conditions are favorable, these sclerotia are close to the host plant root and they can easily germinate causing infection [13].
Currently, the most effective strategies for controlling fungal diseases are resistant cultivars, biological control agents and soil amendments [14-19]. The objective of this study was to evaluate the effect of solid phosphate sludge amendments on the mycelial growth of Verticillium dahliae and Rhizoctonia solani on the PDA based-medium and evaluate the effect of this phosphate sludge on disease development and disease index of tomato and Faba bean plants.
2. MATERIALS AND METHODS
2.1. Origin of the Solid Phosphate Sludge and Soil-borne Pathogens
The solid phosphate sludge was collected from the Khouribga phosphate treatment sludge disposal site. The microorganisms studied have been isolated and identified in the laboratory of Plant Pathology and Postharvest Quality of INRA-Kenitra, Morocco.
2.2. In vitro Inhibition Effect of Solid Phosphate Sludge on Mycelial Growth of Verticillium Dahliae and Rhizoctonia Solani
Solid phosphate sludge at concentrations of 0, 10, 30, 50 and 100 g/l was tested for its effect on mycelial growth of Verticillium dahliae and Rhizoctonia solani and compared to the substrate used by nurseries (Maamora forest soil). Different quantities of sterilized sludge were poured into Erlenmeyer flasks containing the sterilized PDA medium. After stirring, the mixture was dispensed into sterile Petri dishes (9 cm in diameter). Petri dishes containing the substrate-free PDA were used as control. All plates were inoculated centrally with 5 mm diameter discs taken from a 7 days old culture of each soil-borne pathogen. The inoculated Petri dishes were incubated at 25°C ± 2°C. The average diameter growth was calculated of the tested fungi every day until the control reached full growth. The effectiveness of phosphate sludge on mycelial fungal growth was calculated by determining the area in cm2 of mycelial growth and the percentage of inhibition for each pathogen according to the following relation:
In% = (Diameter control – Diameter [C] / Diameter control) x 100, where:
In%: percentage of inhibition,
diameter of the control: diameter of mycelial growth in control dishes,
Diameter [C]: diameter of mycelial growth in dishes with different concentrations of soil amendment.
2.3. In vivo Inhibition Effect of Solid Phosphate Sludge on Verticillium Dahliae and Rhizoctonia Solani
2.3.1. Preparation of Potting Mixes
For the in vivo test with tomato and Faba bean plants, five potting substrates were prepared. Mixed substrates consisted of Maamora forest soil and solid phosphate sludge. The compositions of the mixtures were mixed as volume per volume (v/v) (Table 1).
| Substrates | Composition |
|---|---|
| S1 | 100% of sterile Maamora forest soil |
| S2 | 99% of sterile Maamora forest soil and 1% of sterile phosphate sludge |
| S3 | 97% of sterile Maamora forest soil and 3% of sterile phosphate sludge |
| S4 | 95% of sterile Maamora forest soil and 5% of sterile phosphate sludge |
| S5 | 90% of sterile Maamora forest soil and 10% of sterile phosphate sludge |
2.3.2. Evaluation of the in vivo Inhibition Effect of Solid Phosphate Sludge on Verticillium Dahliae Wilt of Tomato
Two varieties of tomato (Saint pierre and Campbell 33) were used in this experiment. Seeds were sown in peat and transplanted into pots, each containing approximately 1.2 kg potting mix. The pots were placed in a controlled conditions growth greenhouse. Plants were arranged in a completely randomized design with 10 replicates (plants) per treatment. The plants were watered every second day individually and rotated within the growth room (greenhouse) before watering. 30 days after seedling, plant growth was evaluated by measuring shoot fresh and dry weight with a precision analytical balance.
The in vivo inhibition effect of solid phosphate sludge on Verticillium dahliae wilt of tomato was evaluated by pathogenicity experiments at the four-leaf stage performed on tomato plants. Tomato plants were grown under the same conditions in S1, S2, S3, S4 and S5 mixes. The fungal isolates of Verticillium dahliae were transferred to PDA medium (potato dextrose agar) and incubated at 25°C ± 2°C for 7 days. For the pathogenicity experiments, conidia were prepared by transferring pieces of 5 mm diameter, taken from a 7-day old culture on PDA in sucrose sodium nitrate (SSN) [20] in Erlenmeyer flasks and incubated in the dark at 150 rpm for 5 days at 22°C in an orbital shaker. Suspensions before being applied to the plants were centrifuged at 10,000 x g, 12 °C for 10 min, and resuspended in Sterile Distilled Water (SDW) [21].
10 ml conidial suspension of Verticillium dahliae containing 107 conidia ml-1 SDW was inoculated into tomato plants by root drenching. Tomato control plants were also inoculated. The number of leaves showing typical symptoms as a percentage of the total number of leaves of each plant determined disease severity. Periodically for 31 days post-inoculation (dpi), symptoms were recorded for Verticillium dahliae. Disease progression curves were generated over time by the disease assessment. AUDPC (area under the disease progress curve) was calculated by the trapezoidal integration method [21, 22]. The experiment was repeated three times.
2.3.3. Evaluation of the in vivo Inhibition Effect of Solid Phosphate Sludge on Rhizoctonia Solani Disease of Faba Bean
Seeds of two Faba bean (Vica faba L.) varieties (brown and white) were surface-sterilized by rinsing with 70% (v/v) ethanol followed by soaking for 15 min in 3% (v/v) sodium hypochlorite and three drops of Tween- 20 (0.05%), and then rinsed three times with sterile distilled water. The seeds were pre-germinated for 2 days at 25 °C. One seed was planted to a depth of 2 cm in a pot each containing approximately 1.2 kg potting mix. Plants were arranged in a completely randomized design with 10 replicates (plants) per treatment. The plants were watered individually every second day and rotated within the growth room (greenhouse) before watering. 30 days after seedling, plant growth was evaluated by measuring shoot fresh and dry weight with a precision analytical balance.
The inoculum of Rhizoctonia solani was prepared according to the method described by McDonald and Rovira [23]. Autoclaved rice seeds were used. They were placed evenly on Petri dishes surface containing potato dextrose agar, and then a piece of mycelium (5 mm) from a 7-day old culture was placed in the center of the Petri dish. The cultures were incubated at 20°C in darkness for 7 days, and colonized rice seeds were used as inoculum. Six rice seeds were placed at a depth of 5 cm around each plant in the potting mix. In this experiment, the inoculation was carried out at the time of planting. One plant was placed in each pot, with ten replicate pots for each treatment, and the plants were harvested 6 weeks after planting. At harvest, shoots and roots were separated, roots were thoroughly washed and blotted and fresh weights were recorded. To assess the disease severity, the roots were rated visually using a 0–4 scale in which 0 = no lesions, 1 = small lesions, each 1–3 mm, 2 = more extensive lesions, up to 5 mm, 3 = lesions longer than 5 mm and with necrotic areas, 4 = lesions over 50% of the root area and necrosis [24]. A Disease Index (DI) was calculated using the following equation:
% DI =Ʃ (n × Sn) × 100/4N × 100, where:
n = number of plants with a given disease rating, Sn = disease rating, and N = total number of plants rated [24, 25].
2.3.4. Statistical Analysis Method
Data were analyzed by variance analysis (ANOVA), with each data representing the average of three replicates. Thus the data belonging to the same group were considered not different along with the risk equal to 5%. The statistical analysis of the results was carried out using the statistical software.
3. RESULTS
3.1. In vitro Effect of Solid Phosphate Sludge on Mycelial Growth of Verticillium Dahliae
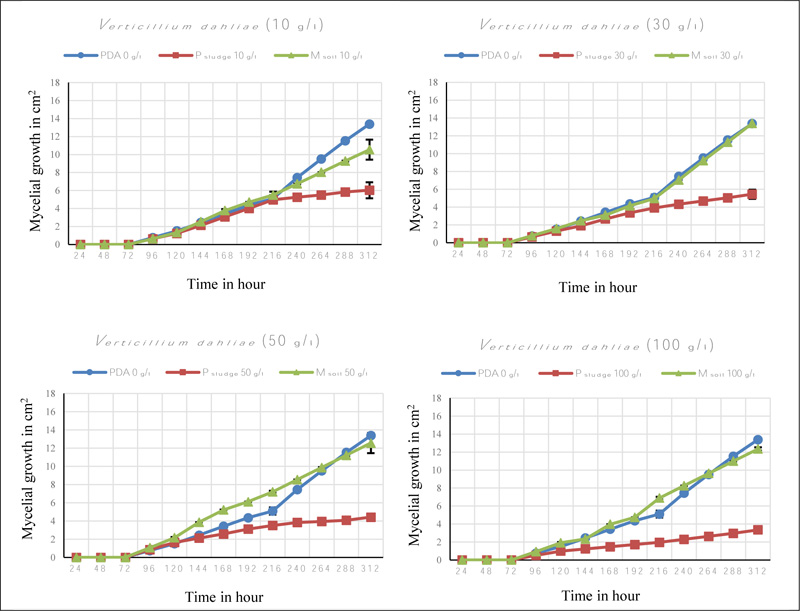
The mycelial growth of Verticillium dahliae was compared in three PDA-based media. The reduction in mycelial growth was observed when the fungus was grown in PDA medium plus 10 g/l of the phosphate sludge compared to the control medium (Figs. 1 and 2).
The mycelial growth decreased with the increase of solid phosphate sludge quantities and the highest reduction of mycelial growth was noted at a concentration of 100 g/l with 4 cm2. This reduction was significantly different to the control medium (p < 0.05).
In addition, no clear difference was observed with the addition of Maamora soil concentrations compared to control medium PDA (Fig. 1).
3.2. In vitro Effect of Solid Phosphate Sludge on Mycelial Growth of Rhizoctonia Solani
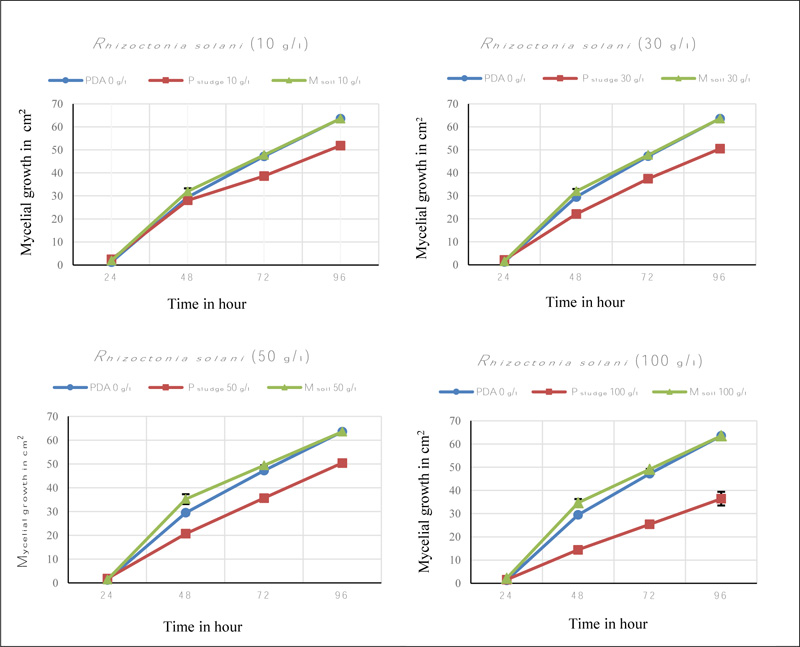
The mycelial growth was significantly affected with the addition of solid phosphate sludge concentrations. This effect is shown in Figs. (2 and 3). The reduction of mycelial growth was greater in solid phosphate sludge concentration of 100 g/l with 40 cm2 that appeared after 48h of incubation and increased with time to significantly differ from control medium PDA (p <0.05).
The effect of Maamora soil on the mycelial growth was not evident and similar to the control medium (PDA only) in all concentrations (Figs. 2 and 3).
3.3. In vitro Inhibition effect of solid phosphate sludge on mycelial growth of Verticillium dahliae and Rhizoctonia Solani
The inhibitory effect of mycelial growth in the three PDA-based media varied dramatically among different concentrations of solid phosphate sludge (Table 2). The inhibition percentage was significantly high for both pathogens Verticillium dahliae and Rhizoctonia solani at solid phosphate sludge concentrations of 100 g/l with 75% and 42% respectively, compared to control medium. In contrast, no evident inhibitory effect was observed in Maamora soil concentrations for Rhizoctonia solani. Otherwise, an inhibition was noted in Maamora soil concentrations of 10 g/l, 50 g/l and 100 g/l but not at 30 g/l for Verticillium dahliae.
| Medium | Concentrations | Rhizoctonia solani | Verticillium dahliae |
|---|---|---|---|
| Inhibition (%) | Inhibition (%) | ||
| PDA + P sludge | 10 g/l | 18.46 b | 55.07 b |
| 30 g/l | 20,60 b | 59.40 b | |
| 50 g/l | 20,71 b | 66,94 b | |
| 100 g/l | 42,63 a | 75,07 a | |
| PDA + M soil | 10 g/l | 0,00 c | 21,34 c |
| 30 g/l | 0,00 c | 0,00 d | |
| 50 g/l | 0,00 c | 6,34 d | |
| 100 g/l | 0,00 c | 7,91 d |
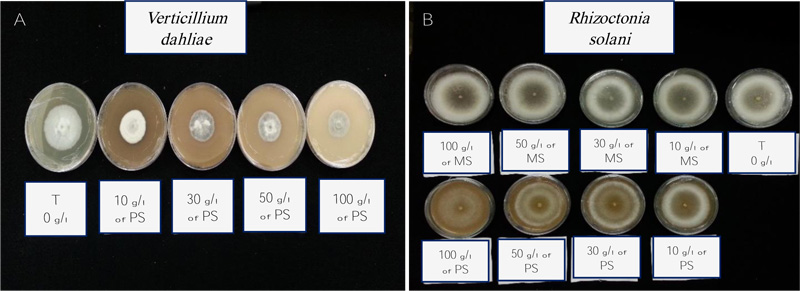
3.4. Evaluation of the in vivo Inhibition Effect of Solid Phosphate Sludge on Verticillium Dahliae Wilt of Tomato
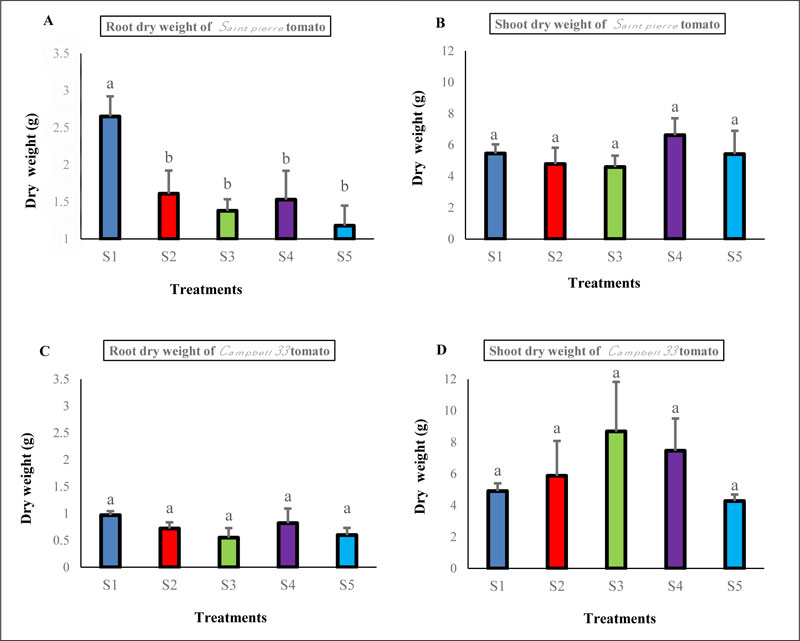
At harvest, Plants grown in Maamora soil substrate visibly stimulated increases in root dry weight for Saint pierre tomato plants but no difference was observed for Campbell 33 tomato plants (Fig. 4A, C) compared to the plants grown in the Maamora soil plus solid phosphate sludge amendment substrates. When plants were grown in the S4 and S3 mixes, an increase in shoot dry weight was observed for Saint pierre tomato and Campbell 33 tomato respectively, albeit the extent of the increase was not significant compared to the other mixes (Fig. 4B, D). The effect of the solid phosphate sludge for potential fungi suppression was evaluated in pathogenicity experiments using tomato plants against Verticillium dahliae.
The plant responses were recorded by the wilting symptoms induced by the pathogens. In this study, tomato plants were grown in substrates based on phosphate sludge concentrations to check whether the doses of the sludge were associated with suppressiveness capacity.
The wilting and yellowing symptoms of Verticillium dahliae appeared especially on older leaves on the 11th day and were recorded until 31 dpi (Fig. 5E, F). Disease (in the form of wilting followed by yellowing and necrosis of leaves) progressed faster in the plants grown in the Maamora soil only, while the plants in Maamora soil and phosphate sludge mix substrates showed less prominent symptoms and slower disease development for Saint pierre and Campbell 33 tomato (Fig. 5A, C). After 31 dpi, disease severity in the S1 plants was 52% and 8% in the S5 plants for Saint pierre tomato (Fig. 4A), whereas in the S1 plants, disease severity was 38.2% (Fig. 5C) and 5% in the S5 plants for Campbell 33 tomato. The AUDPC noted over 31 days of disease progression was higher in the S1 plants than in the other mixes for both varieties (Fig. 5B, D). Similarly, the plants grown in the S5 with 100 g/kg showed less severe symptoms and disease development compared to the plants grown in other mixes (Fig. 5A, C).

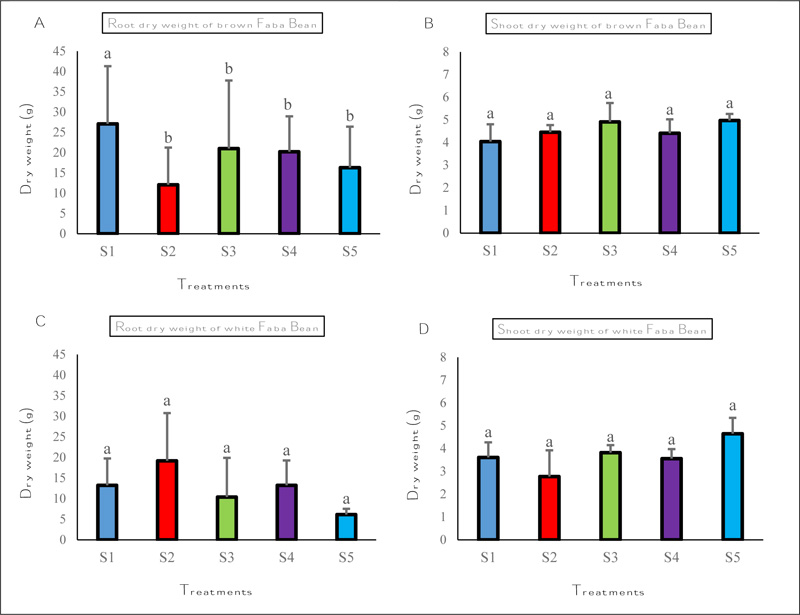
3.5. Evaluation of the in vivo Inhibition Effect of Solid Phosphate Sludge on Rhizoctonia Solani of Faba Bean
At harvest, root dry weight of plants grown in S1 was greater than other plants for brown Faba bean and the extent of the increase was not significant in all mixes for white Faba bean (Fig. 6A, C).
Adding phosphate sludge concentrations had no significant effect on shoot dry weight for brown and white Faba bean.
White Faba bean (Fig. 6A, C). Adding phosphate sludge concentrations had no significant effect on shoot dry weight for brown and white Faba bean.
S1 (Maamora soil only) plants inoculated with Rhizoctonia solani showed higher disease ratings with 75% and 72.5% for brown and white Faba bean respectively, than other mixes S2, S3, S4 and S5. The less severe symptoms were recorded in the S5 mix plants with 100 g/kg of phosphate sludge with 45% and 35% disease index for brown and white Faba bean respectively (Table 3).
| Treatments | Brown Faba bean | White Faba bean | ||
|---|---|---|---|---|
| Disease rating (0–4) * | Disease index (%)** | Disease rating (0–4)* | Disease index (%)** | |
| S1 | 3 | 75 | 2,9 | 72,50 |
| S2 | 3 | 75 | 2,7 | 67,50 |
| S3 | 2,9 | 72,50 | 2,3 | 57,50 |
| S4 | 2,7 | 67,50 | 2,1 | 52,50 |
| S5 | 1,8 | 45 | 1,4 | 35 |
** Disease Index was calculated from Ʃ (number of plants of a given disease rating × disease rating) × 100/ 4 (total number of plants rated) × 100.
4. DISCUSSION
Recently, the search for alternative methods to control plant diseases has aimed at eliminating the negative impact of the approaches currently used, such as synthetic fungicides and resistant cultivars, on public health and the environment. There is a substantial number of studies demonstrating the effective control of soil-borne pathogens by the application of composts and the disease suppression conferred by soil amendments or composts has been systematically reviewed [21].
However, the accumulation of these phosphate sludges in the environment is a serious problem in terms of storage capacities in the country. They modify topography, reduce arable lands, and disfigure the landscapes. In addition, during open pit mining, top soils are lost because they are mixed with other materials in waste rocks and overburden piles since no waste sorting is implemented [26]. Thus, the recycling and recover part of the phosphate and other nutrients could be a useful alternative to limit their negative impacts.
In the present study, our first goal was to evaluate the potential suppressive properties of solid phosphate sludge amendments in vitro and in vivo against the main fungal pathogens of tomato and Faba bean and then to evaluate the effect of amendment application on root and shoot dry weights of these plants. The inhibitory effect of mycelial growth was observed in high concentrations of phosphate sludge-amended medium compared to Maamora soil and PDA only. Disease suppressiveness was studied in solid phosphate sludge amendment under controlled environmental conditions and although pathogens were inoculated at high densities, the tomato and the Faba bean plants, grown in the high concentrations of phosphate sludge showed slower disease development and less severe symptoms compared to the control plants for both pathogens. Additionally, the application of the phosphate sludge did not affect visibly and quantitatively the dry weight of tomato and Faba bean plant varieties compared to the control.
Our study demonstrated that solid phosphate sludge amendments were suppressive against diseases of tomato and Faba bean; the second goal was to show that suppressiveness was due to differences in the chemical composition of the substrates used. The solid phosphate sludge is mainly composed of clay, and the contents of total limestone and active limestone are high and the pH is alkaline due to the high contents of limestone.
The contents of mineral nitrogen and exchangeable potassium are very low, the total phosphorus content is slightly elevated and the amounts of zinc and cadmium are considerably high.
Rowena Sjaan Davey [27] suggests that in highly calcareous soils like solid phosphate sludge, the fixation of phosphorus decreased plant growth can limit the soil microbial populations and is likely to decrease the soil microbial population growth and activity resulting in a decrease in general disease suppression. In addition, Rowena also showed that the addition of orthophosphate in a highly calcareous soil increased the amount of pathogen (detected in the soil as Rhizoctonia solani DNA) and severity of Rhizoctonia root-rot, as assessed from root symptoms, but did not affect the virulence of the pathogen [27].
Additionally, Abeer M. Mousa [28] showed that there is an effect of superphosphate (0, 100, 200 Kg/fed) on the percentage of pre- and post-emergence damping-off, root-rot disease severity caused by the fungal pathogens, Rhizoctonia solani. Abeer also reported that all tested phosphorus fertilizers significantly reduced the development of the root-rot disease.
Heavy metals have toxic effects on soil microbiota and are known as inhibitors of enzyme activities [29-31]. This behavior was observed in Finland with zinc and cadmium contaminated soil where the fungi decreased with metal contamination [32]. Cadmium had a negative effect on soil micro-organisms count in inhibiting fungi [33]. Babich and Stotzky [34, 35] grouped microscopic fungi into three Cd-sensitivity categories. The first category consisted of fungi capable of growth in the presence of up to 10 μg of Cd/ml but are inhibited by 100 μg of Cd/ml. The second category consisted of fungi capable of growth in the presence of up to 100 μg of Cd/ml but are inhibited by 1000 μg of Cd/ml while the third category comprised fungi growing in the presence of above 1000 μg of Cd/ml [36]. In comparison with the above-quoted studies, the solid phosphate sludge containing 33 000 μg of Cd/ml [26] can be responsible for this inhibition and disease severity and index reduction for the both pathogens.
In the present study, it was interesting to note that, after physicochemical analyzes, the phosphate sludge contained high concentrations of Zn (3.6 to 4.7 mg/kg). Siddiqui [37] showed that, the Zn at high concentrations (0.8 and 1.6 mg/kg soil) caused significant suppression of Rhizoctonia solani. In another study, Streeter [38] demonstrated that the application of Zn did not directly inhibit infection by Rhizoctonia solani, nor reduced its pathogenicity, but it strongly increased root growth. Consequently, we suggest that the high concentration of Zn in phosphatic sludge is the main cause for Rhizoctonia solani growth inhibition.
Furthermore, there was an inhibition in mycelia growth for Verticillium dahliae in different concentrations of solid phosphate sludge and the disease severity in tomato plants. These results were confirmed by Davis [39] who showed that there was a significant effect of phosphate levels upon disease suppression. As phosphate concentrations in the soil increased to the optimum, the colonization of Verticillium dahliae in potato stem-tissue decreased. Davis [40] also demonstrated that adequate N and P fertilization can reduce Verticillium wilt of Russet Burbank potato and minimize yield losses that commonly occur with the intensive cropping of potato and reported suppression of Verticillium dahliae with high rates of N and P. However, working on a soil with very high phosphate levels, Platt and Arsenault [41] saw no effect of additional phosphate up to 280 kg P ha−1 on either Verticillium dahliae or Verticillium albo-atrum. Arsenault also demonstrated that suppression of Verticillium wilt caused by Verticillium dahliae did not affect variedly in applied rates of nitrogen or phosphorus [41].
Moreover, other works have reported suppressive effects of phosphites on oomycete growth [42-45]. Borza [46] reported that the benefits of using phosphites extend beyond oomycetes pathogens, as they can limit the development of pathogenic Verticillium species. Adequate P levels can suppress Verticillium wilt in potato plants [47]. However, Romania [47] showed in their study that increasing P availability did not reduce the incidence of Verticillium wilt.
Several physicochemical parameters of the soil, such as soil pH, phosphorus and other elements have been often related to disease suppression [48]. Other studies suggest that fungal distributions are more influenced by N and P availability than pH per se [49, 50]. Otherwise, for the fungi at least, it appears that most phosphatases released for extracellular phosphate acquisition have acidic pH optima, whereas those intended for intracellular reactions have optima at neutral to alkaline pH [51]. Inositol phosphates, produced mostly as phosphate storage products by plants, account for half of soil organic phosphate [52, 53]. So it can be suggested that alkaline pH of phosphate sludge is at disadvantage as the fungi enzymes for phosphorus degradation and acquisition associate with other nutrients as nitrogen and carbon.
For a large number of plants, it has been indicated that the optimal concentration of phosphate in the soil is 1-10 μM, whereas 10-20 μM in the cytoplasm of the plant cell. Compared with higher plants, the fungus requires a large amount of phosphate [54, 55]. But, Zenichi Moromiza [56] suggests that a large quantity of phosphorus is not always necessary for the hyphal development, but indispensable for the sclerotium initiation and subsequent development, enlargement and maturation which are required for the high energy production.
CONCLUSION
In this study, phosphate sludge was used as a basic composition to develop combinations of substrate for plant cultivation. According to our results, the phosphate sludge has a great potential to suppress Verticillium dahliae and Rhizoctonia solani. Furthermore, with this study, we have been able to use this sludge or incorporate into substrates in the nurseries to be used as an amendment soil to increase the performance of soil against soil-borne pathogens and protect production systems. In order to recommend its use where Rhizoctonia solani and Verticillium dahliae are limiting factors, more work is needed to understand the phenomenon. Additional investigations should also be undertaken to determine the effect of solid phosphate sludge on Verticillium dahliae and Rhizoctonia solani in the field.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No human or animals were used in this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
This work was conducted under a project sponsored by OCP Foundation under the number BIO-ELG-01/2017.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


